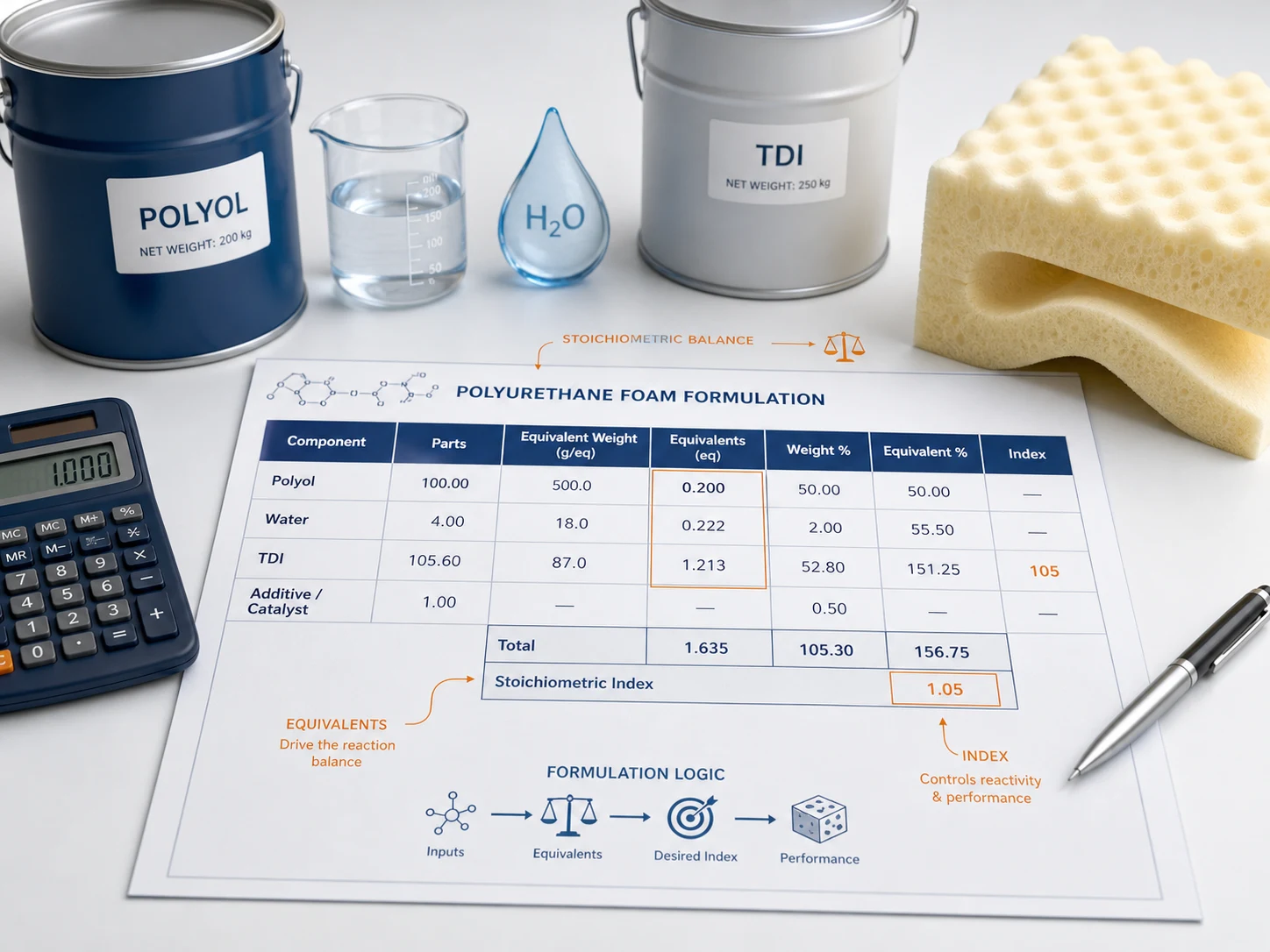
Introduction
A polyurethane formulation sheet looks like a recipe.
Component names are listed in one column. Quantities are written in another. A target index may appear at the bottom. At first glance, it looks like a simple list of raw materials.
But a good polyurethane formulation sheet is much more than a recipe. It is a stoichiometric document.
Every reactive component connects to every other reactive component through equivalent weight, reactive equivalents, and the isocyanate index. The sheet does not only tell you what goes into the foam. It tells you whether the chemistry is balanced.
Most people read only part of the sheet.
Operators often read the parts column. Buyers usually read component names and quantities. Engineers may check the index, but sometimes ignore equivalent percentages or whether the equivalent weights still match current raw material data.
That is risky.
A polyurethane formulation sheet contains enough information to verify whether the formula is chemically consistent — but only if every column is read correctly.
This guide explains what each column means, how reactive and non-reactive components should be read differently, and why the index line is a verification tool, not just a target number.
What a Polyurethane Formulation Sheet Actually Tells You
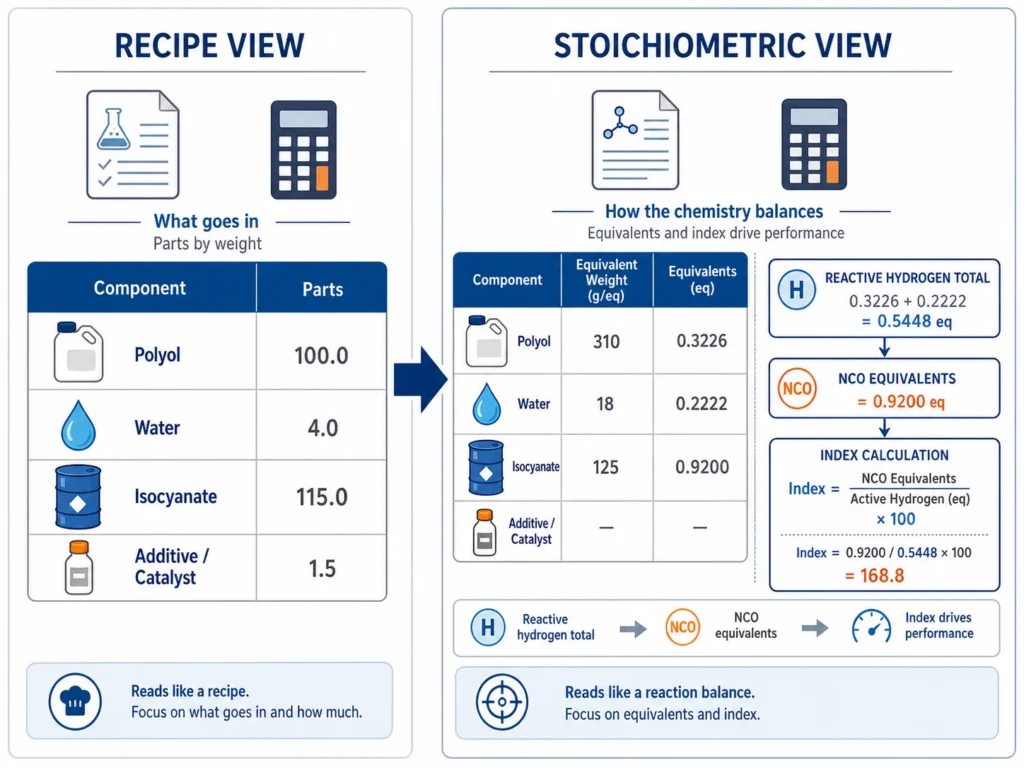
A polyurethane formulation sheet tells you two different things at the same time.
First, it tells you the recipe — component names, parts by weight, catalyst levels, surfactant levels, isocyanate quantity, target index.
Second, it tells you the stoichiometry — equivalent weight, reactive equivalents, total reactive hydrogen equivalents, NCO equivalents, actual index, and chemical balance between components.
The recipe tells production what to add. The stoichiometry tells the engineer whether the formula makes chemical sense.
That difference is important. A formula can look correct as a recipe and still be wrong as a stoichiometric calculation.
For example, a sheet may show the correct component names and familiar parts. But if water equivalent weight is entered incorrectly, or if the isocyanate %NCO value is outdated, the index calculation may be wrong.
The formula sheet looks normal. The foam chemistry is not normal.
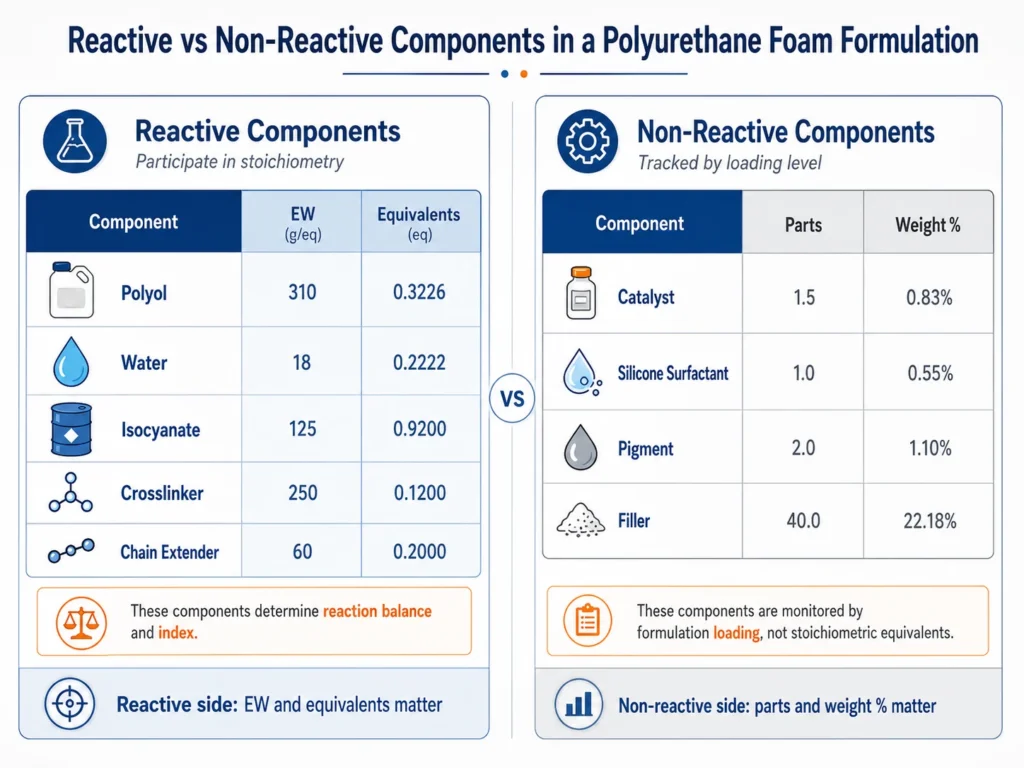
Reactive and Non-Reactive Components Are Not Read the Same Way
The first step in reading a polyurethane formulation sheet is separating reactive components from non-reactive components.
Reactive components participate directly in the stoichiometric calculation. They include polyol, water, isocyanate, crosslinkers, chain extenders, and amine-functional reactive additives. Reactive components need equivalent weight and equivalent calculations.
Non-reactive components do not normally appear in the index calculation. They include catalysts, silicone surfactants, some flame retardants, pigments, fillers, and processing aids.
Non-reactive components still matter — they affect reaction speed, cell structure, processing, surface quality, and sometimes regulatory or cost calculations. But they do not contribute reactive equivalents to the isocyanate index.
This means they should not be read the same way as reactive components. A catalyst at 0.30 parts may strongly affect cream time and rise profile, but it does not have an equivalent weight in the index calculation. A surfactant at 1.00 part may control cell stability, but it does not determine stoichiometric balance.
Reactive components define the chemistry. Non-reactive components control how the chemistry behaves in production.
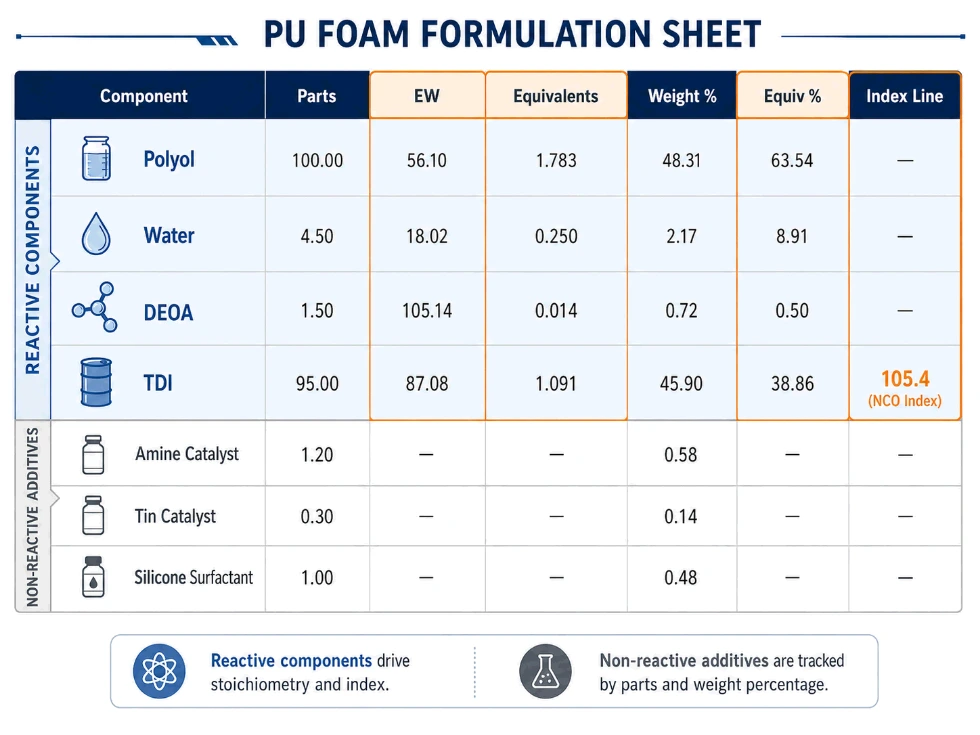
Example Polyurethane Formulation Sheet
Below is a simplified flexible polyurethane foam formulation example.
| Component | Parts | EW | Equivalents | Weight % | Equiv % |
|---|---|---|---|---|---|
| Polyol, OHV 51 | 100.00 | 1,100 | 0.09091 | 64.05% | 16.54% |
| Water | 4.00 | 9 | 0.44444 | 2.56% | 80.86% |
| DEOA crosslinker | 0.50 | 35.0 | 0.01429 | 0.32% | 2.60% |
| Total reactive H | 0.54964 | 100% | |||
| Amine catalyst | 0.30 | — | — | 0.19% | — |
| Tin catalyst | 0.15 | — | — | 0.10% | — |
| Silicone surfactant | 1.00 | — | — | 0.64% | — |
| TDI 80/20, 48.3% NCO | 50.17 | 86.96 | 0.57695 | 32.14% | — |
| Total formula | 156.12 | 100% |
Index calculation:
Index = NCO equivalents ÷ Total reactive H equivalents × 100
Index = 0.57695 ÷ 0.54964 × 100 = 105.0
This table contains five important numbers for each reactive component:
- Parts
- Equivalent weight
- Equivalents
- Weight percentage
- Equivalent percentage
The index line then verifies whether the reactive balance matches the stated target.
Column 1: Parts by Weight
Parts by weight are usually expressed relative to 100 parts of polyol. This is the standard convention in PU foam formulation.
For example: Polyol = 100 parts, Water = 4 parts, DEOA = 0.5 parts, TDI = 50.17 parts.
This allows formulas to be scaled easily. If production needs a larger batch, the ratio can be increased while keeping the same formula structure.
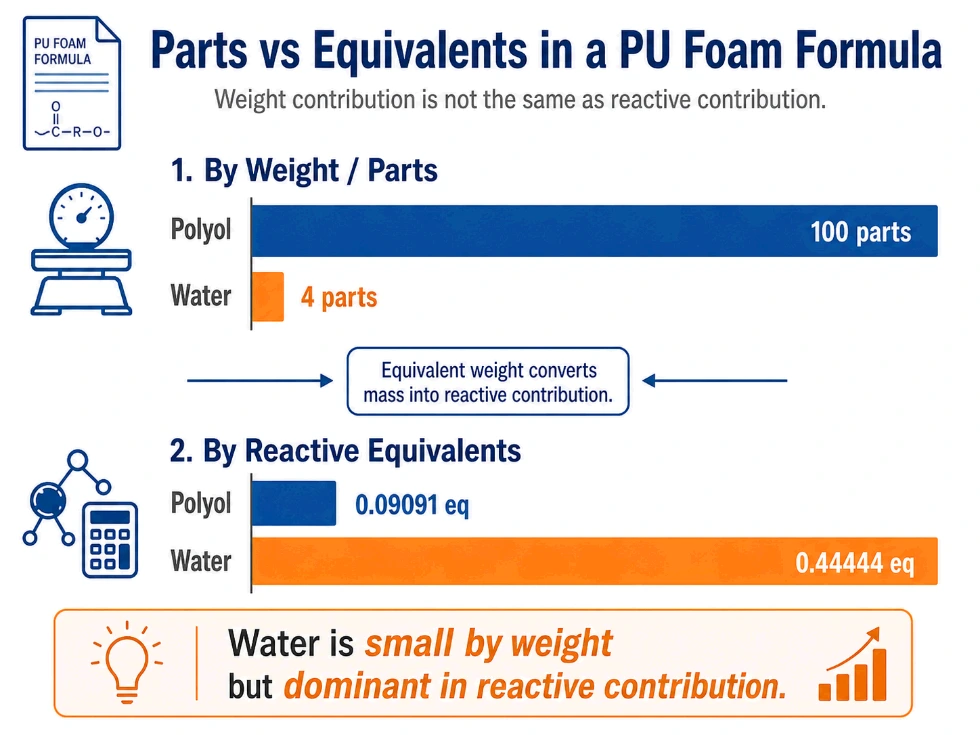
Parts are useful for production. But parts alone do not tell you reactivity. This is a major mistake.
At first glance, 100 parts polyol and 4 parts water look like the polyol dominates the formula. By mass, it does. But chemically, water contributes far more reactive equivalents because its equivalent weight is much lower.
So the parts column tells you how much material is added. It does not tell you how much reactive chemistry that material contributes.
Column 2: Equivalent Weight
Equivalent weight converts mass into reactivity. It tells you how many grams of a component contain one equivalent of reactive groups.
Each reactive component has its own equivalent weight. Typical calculations include:
Polyol EW = 56,100 ÷ OHV → For OHV 51: Polyol EW = 56,100 ÷ 51 = 1,100
Water EW = 18 ÷ 2 = 9
DEOA EW = Molecular Weight ÷ Reactive Groups → DEOA EW = 105.14 ÷ 3 = 35.0
TDI EW = 4,200 ÷ %NCO → For TDI at 48.3% NCO: TDI EW = 4,200 ÷ 48.3 = 86.96
Equivalent weight is one of the most important columns in the sheet. If the EW value is wrong, every downstream calculation becomes wrong.
A formulation sheet should never use outdated equivalent weights copied from old raw material data.

Column 3: Equivalents
The equivalents column is where the formula becomes chemically readable.
The calculation is:
Equivalents = Parts ÷ Equivalent Weight
This converts every reactive component onto the same stoichiometric basis.
| Component | Parts | EW | Equivalents |
|---|---|---|---|
| Polyol | 100.00 | 1,100 | 0.09091 |
| Water | 4.00 | 9 | 0.44444 |
| DEOA | 0.50 | 35.0 | 0.01429 |
| TDI | 50.17 | 86.96 | 0.57695 |
This column reveals something that the parts column hides.
- Water is only 4 parts, but contributes 0.44444 reactive equivalents.
- Polyol is 100 parts, but contributes only 0.09091 reactive equivalents.
That means water is a small component by weight but a dominant reactive component in the formula.
This is why water level is so powerful in flexible foam formulation. A small water change can move the chemistry more than the parts column suggests.
Column 4: Weight Percentage
Weight percentage tells you how much each component contributes to the total formula mass.
The calculation is:
Weight % = Component parts ÷ Total formula parts × 100
In the example formula, total formula weight is 156.12 parts.
- Polyol weight % = 100 ÷ 156.12 × 100 = 64.05%
- Water weight % = 4 ÷ 156.12 × 100 = 2.56%
- TDI weight % = 50.17 ÷ 156.12 × 100 = 32.14%
Weight percentage matters for cost calculation, raw material usage, regulatory limits, flame retardant loading, VOC review, production costing, and procurement analysis.
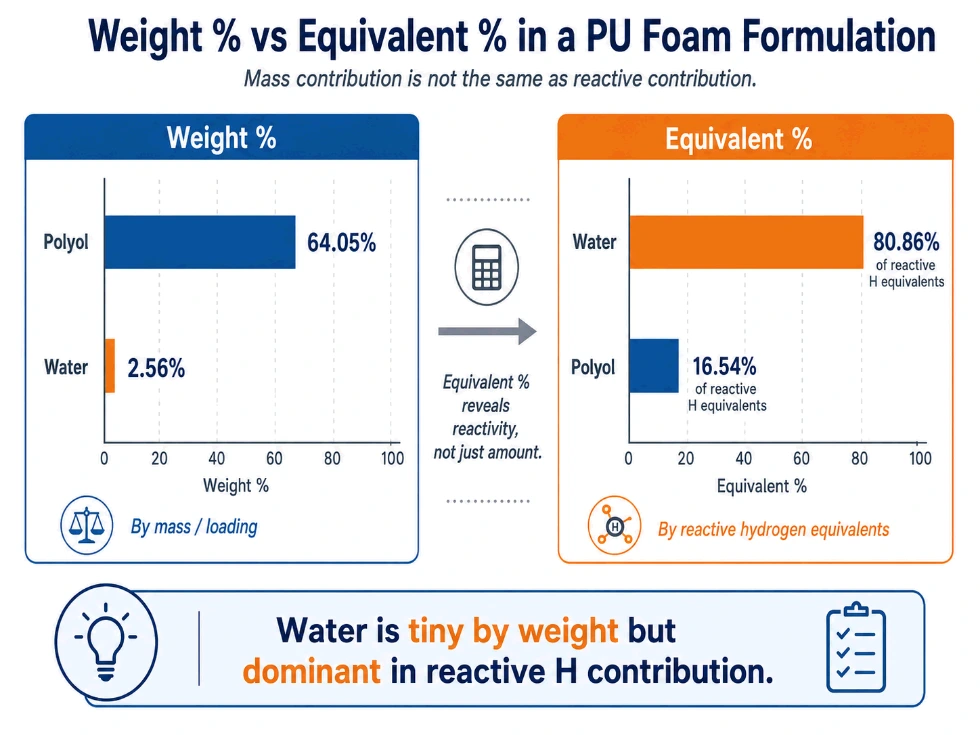
Weight percentage is important, but it should not be confused with reactive contribution. A component can have low weight percentage and high equivalent percentage. Water is the clearest example.
Column 5: Equivalent Percentage
Equivalent percentage shows each reactive component’s share of total reactive hydrogen equivalents.
For reactive hydrogen components:
Equivalent % = Component equivalents ÷ Total reactive H equivalents × 100
| Component | Equivalents | Equivalent % |
|---|---|---|
| Polyol | 0.09091 | 16.54% |
| Water | 0.44444 | 80.86% |
| DEOA | 0.01429 | 2.60% |
| Total | 0.54964 | 100% |
This is one of the most important views in the sheet. It shows which components dominate the reactive chemistry.
In this example, water is only 2.56% by weight, but it contributes 80.86% of the reactive hydrogen equivalents.
That is why water adjustments are powerful. The equivalent percentage column helps engineers see the formula chemically, not just by mass.
The Index Line: Verification, Not Just a Target
The isocyanate index is calculated from the equivalents column.
Index = NCO equivalents ÷ Total reactive H equivalents × 100
Using the example: Index = 0.57695 ÷ 0.54964 × 100 = 105.0
This line verifies whether the formula is chemically balanced at the intended index.
If the sheet states Index 105 but the equivalents calculate to another value, the formula contains an error. Possible causes include:
- Wrong polyol EW
- Wrong water EW
- Wrong DEOA EW
- Wrong TDI %NCO
- Outdated CoA values
- Incorrect parts
- Copied formula values
- Manual spreadsheet error
The index line should not be trusted blindly. It should be recalculated.
A good formulation sheet self-verifies when every column is correct.
How to Read Non-Reactive Components
Non-reactive components are read differently. Catalysts and surfactants do not usually contribute equivalents to the index calculation.
| Component | Parts | Function |
|---|---|---|
| Amine catalyst | 0.30 | Blowing / gelling balance |
| Tin catalyst | 0.15 | Gelling rate and cure |
| Silicone surfactant | 1.00 | Cell nucleation and stability |
These materials are not unimportant. They are extremely important for process control. But they do not fix stoichiometric errors.
A catalyst package can make a wrong formula react faster or slower. It cannot make a wrong index correct. A surfactant can improve cell structure. It cannot correct a missing equivalent calculation.
This is one of the most important formulation-reading rules:
Do not use process additives to fix stoichiometric errors. First verify the reactive calculation. Then tune the process additives.
Practical Checklist for Reading a PU Foam Formulation Sheet
Use this checklist when reviewing a formulation sheet:
| Checkpoint | Question |
|---|---|
| Reactive components | Are polyol, water, crosslinkers, chain extenders, and isocyanate clearly identified? |
| Non-reactive components | Are catalysts, surfactants, and additives separated from index calculation? |
| Parts | Are all parts based on 100 parts polyol? |
| Polyol EW | Is EW calculated from actual OHV? |
| Water EW | Is water entered as 9? |
| Isocyanate EW | Is EW calculated from actual %NCO? |
| Crosslinker EW | Are all reactive groups included? |
| Equivalents | Are parts divided by correct EW? |
| Total reactive H | Is the total correct? |
| Index | Does calculated index match the stated index? |
| Weight % | Does total formula weight equal 100% after conversion? |
| Equivalent % | Does the reactive contribution make chemical sense? |
This checklist turns the sheet from a recipe into a verifiable technical document.
Use the PolymerIQ Calculators
Equivalent weight values must be correct before the formulation sheet can be trusted. The PolymerIQ Equivalent Weight Calculator helps verify EW from OHV and other raw material values. Use it when reviewing a new formula, checking a copied formula, updating polyol OHV, checking crosslinker EW, or auditing old formula sheets.
Open the Equivalent Weight Calculator →
The index line should always be verified from actual equivalents. The PolymerIQ NCO / TDI Index Calculator helps check whether the formula is really running at the intended index. Use it when reviewing a formulation sheet, updating isocyanate %NCO, changing water level, checking CoA values, auditing legacy formulas, or confirming TDI or MDI parts.
Open the NCO / TDI Index Calculator →
For the full equivalent weight guide, read Equivalent Weight in Polyurethane Foam: Complete Calculation Guide.
For water’s role in the formula, read The Dual Role of Water in Polyurethane Foam: Blowing Agent and Urea Network Builder.
For the NCO content explanation, read NCO Content in Isocyanate: What %NCO Means in PU Foam Formulation.
For the next article on formula columns, read Parts, Equivalent Weight, and Equivalents in a PU Foam Formula: What Each Column Means.
For the audit article, read What a Polyurethane Formulation Sheet Cannot Tell You.
FAQs
What does a polyurethane formulation sheet contain?
A polyurethane formulation sheet contains both a recipe (component names, parts by weight, target index) and a stoichiometric calculation (equivalent weight, reactive equivalents, total reactive H, NCO equivalents, actual index). The recipe tells production what to add. The stoichiometry tells the engineer whether the chemistry is balanced. A complete sheet contains both, with one verifying the other.
What’s the difference between reactive and non-reactive components on a formulation sheet?
Reactive components (polyol, water, isocyanate, crosslinkers, chain extenders) participate in the polyurethane reaction and contribute reactive equivalents to the isocyanate index calculation. Non-reactive components (catalysts, silicone surfactants, pigments, fillers) affect process behaviour, cell structure, and properties but do not contribute equivalents. They should be read differently — reactive components need EW and equivalents, non-reactive components need only parts and weight percentage.
Why does a formulation sheet show parts based on 100 parts polyol?
Using 100 parts polyol as the reference base is a standard PU foam convention. It allows easy scaling — production can multiply the formula to any batch size while keeping the same ratios. It also makes formulas easier to compare across products and suppliers since polyol is the largest reactive component by mass.
How do I calculate equivalents from parts in a formula?
Use Equivalents = Parts ÷ Equivalent Weight for each reactive component. For example, 4 parts of water with EW = 9 gives 4 ÷ 9 = 0.44444 equivalents. 100 parts of polyol with EW = 1,100 gives 100 ÷ 1,100 = 0.09091 equivalents. Sum the reactive hydrogen equivalents to get the total used in the index calculation.
Why is water dominant in the equivalent percentage column even though it’s only a few parts by weight?
Water has a very low equivalent weight (9 g/eq) compared to polyol (around 1,000–1,200 g/eq). Even though water makes up only 2–4% of the formula by weight, it contributes a much larger share of total reactive hydrogen equivalents — often 70–85% in flexible foam. This is why a small water change creates a large stoichiometric effect, and why water adjustments are so powerful.
What does the equivalent percentage column tell me?
Equivalent percentage shows each reactive component’s share of total reactive hydrogen equivalents. It reveals which components actually dominate the reactive chemistry, which is often very different from what the parts or weight percentage columns suggest. For example, water can be 2–3% by weight but 80% by equivalent percentage. Reading this column helps engineers see the formula chemically, not just by mass.
Should I trust the index value printed on the formulation sheet?
No — the index line should be recalculated, not trusted blindly. Calculate Index = NCO equivalents ÷ Total reactive H equivalents × 100 and compare with the stated value. If they don’t match, the formula contains an error somewhere — wrong EW, outdated %NCO, missing reactive component, copied values, or a spreadsheet error. The index line is a verification tool, not just a target.
Can I fix a formulation problem by adjusting catalyst or silicone?
Catalysts and silicones are powerful for process control — they affect reaction speed, cell structure, and surface quality. But they cannot fix stoichiometric errors. If the index is wrong because of incorrect EW values, missing reactive components, or outdated %NCO, no amount of catalyst tuning will restore the network the missing or excess NCO would have built. First verify the reactive calculation, then tune the process additives.
What are the most common errors in a polyurethane formulation sheet?
Common errors include: water EW entered as 18 instead of 9, polyol EW based on outdated OHV, isocyanate EW based on TDS midpoint instead of actual CoA %NCO, crosslinker (especially DEOA) calculated from OHV alone without including amine hydrogens, missing reactive components from the index calculation, and copied formula values that no longer match current raw material data. Any one of these can corrupt the entire stoichiometric calculation while the sheet still looks correct.
How often should I audit a formulation sheet?
Audit any time the formula has been adjusted, copied, inherited from another plant, or used for many production runs without verification. Also audit when foam properties drift unexpectedly, when a new supplier or grade is introduced, when CoA values change meaningfully, or when persistent quality problems do not respond to standard process corrections. A formulation sheet that has not been audited in years often contains accumulated errors.
Key Takeaways
A polyurethane formulation sheet is not just a recipe. It is a stoichiometric document.
Reactive and non-reactive components must be read differently:
- Reactive components need equivalent weight, equivalents, and index calculation.
- Non-reactive components affect process, cell structure, and performance, but they do not normally contribute to the index.
For each reactive component, the key numbers are:
- Parts
- Equivalent weight
- Equivalents
- Weight percentage
- Equivalent percentage
The index line then verifies whether the reactive balance matches the intended target.
- Parts show how much material is added.
- Equivalent weight converts mass into reactivity.
- Equivalents show chemical contribution.
- Weight percentage supports cost and compliance.
- Equivalent percentage shows which components dominate the reactive chemistry.
The index line should be recalculated, not trusted blindly.
If a formulation sheet cannot be verified from its own numbers, it should not be treated as production-ready.
Conclusion
If your formulation sheet has been copied, inherited, or adjusted over time, the numbers may no longer reflect the actual chemistry being run in production.
PolymersIQ can help verify every reactive component, every equivalent weight, every equivalent value, and the final index calculation to identify silent formulation errors before they become foam quality problems.
To get accurate support, please share:
- Your current formulation sheet (with EW values)
- Polyol grade, OHV, and supplier
- Isocyanate type and current CoA %NCO
- Water level, crosslinker, and chain extender data
- Target index and any observed foam quality issues
- Description of the problem and adjustments already tried
Contact PolymerIQ for a formulation sheet audit →