Introduction
The dual role of water in PU foam is one of the most important formulation controls in flexible polyurethane systems. Water is one of the smallest ingredients in a flexible polyurethane foam formula, but it is also one of the most chemically active. It does not only generate CO₂ for blowing; it also reacts with isocyanate to form urea hard segments that influence density, hardness, exotherm, cure behavior, and NCO demand.
In a typical flexible foam formula, water may be only 2–4 parts per 100 parts of polyol — a small fraction by weight. But its equivalent weight is just 9 g/eq, far lower than any other reactive component. That low EW makes water dominant in the reactive hydrogen calculation. In many flexible foam formulas, water contributes 70–85% of the reactive hydrogen equivalents while being only 2–3% of the formula by weight.
That alone would make water powerful. But water does something else that changes the chemistry of the foam.
Water has two reactive roles in polyurethane foam, not one:
- It is the chemical blowing agent. Water reacts with isocyanate to generate CO₂ gas, which expands the foam and creates the cellular structure.
- It is a urea network builder. The same reaction also produces an amine intermediate, which reacts again with isocyanate to form urea linkages — hard segments that strengthen the polymer network.
Most production engineers know the first role. Many do not consciously consider the second. That is why water level changes can produce surprising effects on foam hardness, compression set, and exotherm — not just density.
This article explains the chemistry of water’s dual role, why it makes water the most powerful adjustment variable in flexible foam formulation, and what changes when water level moves up or down.
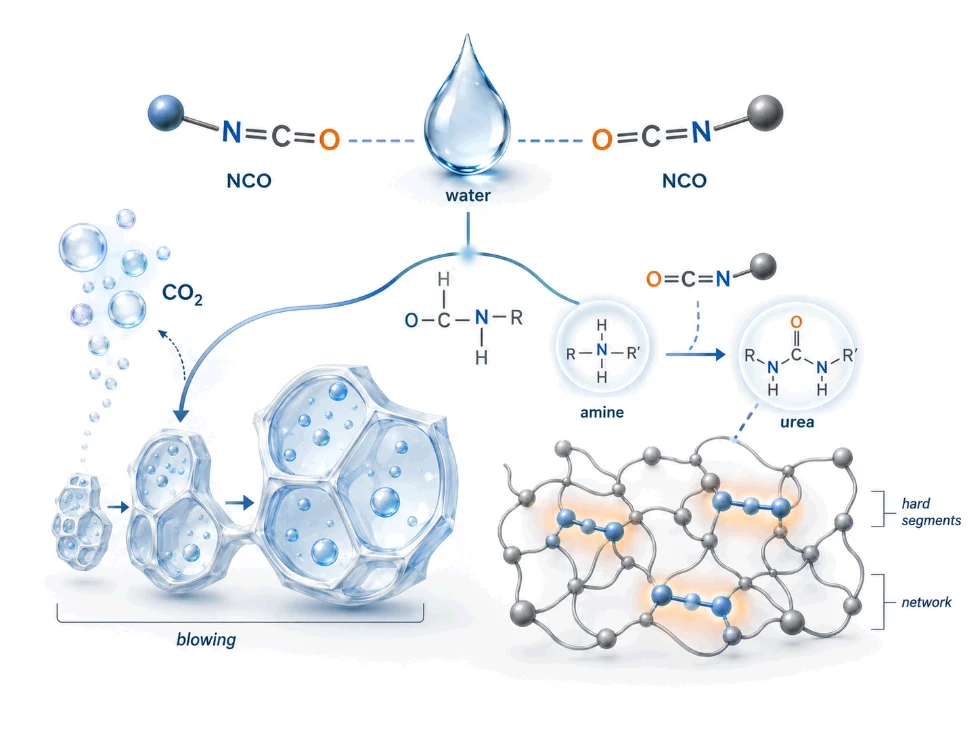
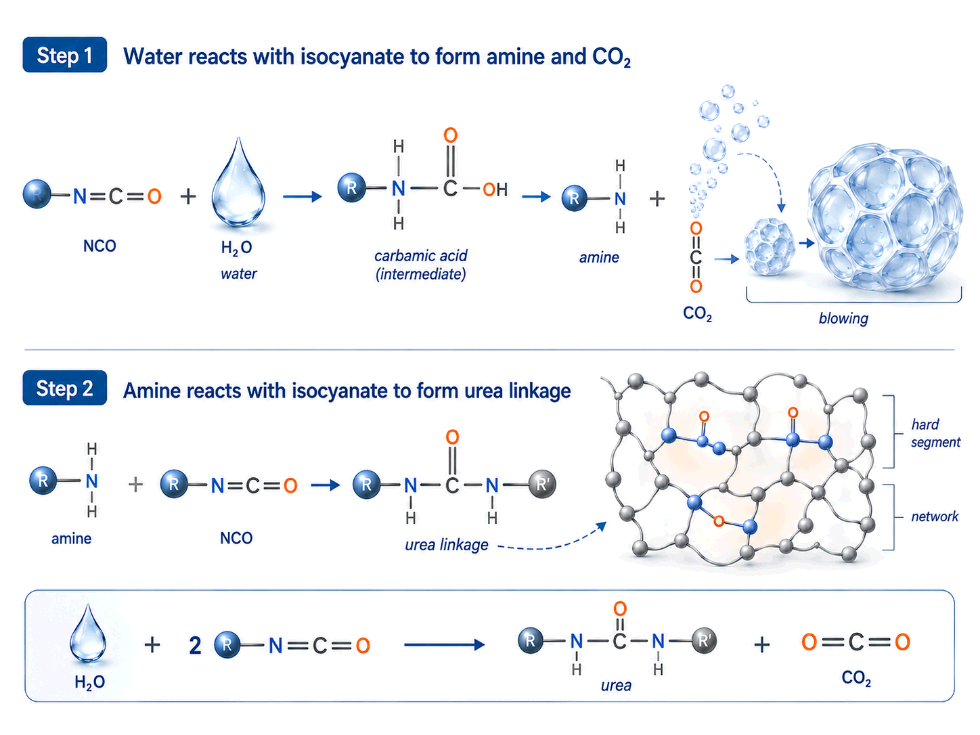
The Two-Step Water Reaction with Isocyanate
Water and isocyanate react in two sequential steps. Both steps consume NCO equivalents, and both contribute to the final foam structure.
Step 1: Water reacts with NCO to form carbamic acid
R-NCO + H₂O → R-NH-COOH (carbamic acid intermediate)
The carbamic acid is unstable. It immediately decomposes:
R-NH-COOH → R-NH₂ + CO₂
This produces an amine and releases CO₂ gas. The CO₂ is the blowing agent that expands the foam. This is the role most engineers know.
Step 2: The amine reacts with another NCO to form urea
R-NH₂ + R’-NCO → R-NH-CO-NH-R’ (urea linkage)
The amine produced in Step 1 is itself reactive. It reacts with a second NCO group to form a urea linkage. This urea is built into the polymer network as a hard segment.
The combined reaction
When the two steps are combined, one water molecule consumes two NCO equivalents:
H₂O + 2 R-NCO → R-NH-CO-NH-R + CO₂
This is why water has two reactive hydrogens for the index calculation. Its molecular weight is 18 g/mol, and it provides 2 reactive equivalents:
Water EW = 18 ÷ 2 = 9 g/eq
The very low equivalent weight comes directly from this two-step chemistry. Water is small, and it consumes NCO at two sites — that is the source of its formulation power.
Why Water Has Two Reactive Hydrogens
A common point of confusion is whether water EW should be 18 (its molecular weight) or 9 (molecular weight divided by 2). The correct value for PU foam stoichiometry is 9.
The reason is that one water molecule reacts with two NCO groups across the two-step reaction. Each H–O–H bond contributes one reactive hydrogen, but in PU foam chemistry, both hydrogens are consumed — first to form the carbamic acid (Step 1), and then through the amine-to-urea reaction (Step 2).
Water molecular weight: 18 g/mol
Reactive hydrogens per water molecule: 2
Water equivalent weight: 18 ÷ 2 = 9 g/eq
Using EW = 18 in the formula sheet is one of the most common errors in PU foam stoichiometry. It under-counts water’s reactive contribution by half, which produces an apparent index that does not match the actual running index.
If the sheet uses EW = 18 instead of 9, the formula appears to have less reactive hydrogen than it actually does. The calculated index reads higher than reality. The foam may then show signs of insufficient NCO — soft cure, weaker hardness, compression set issues — even though the formula sheet says everything is correct.
Always use Water EW = 9 in flexible PU foam stoichiometry.
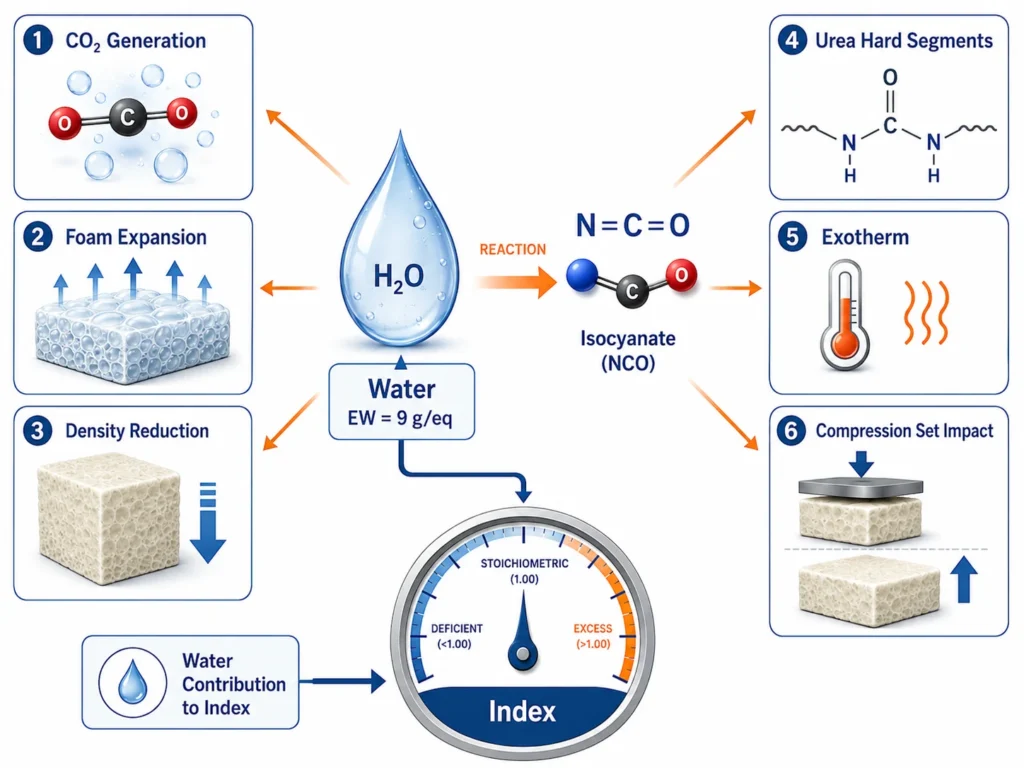
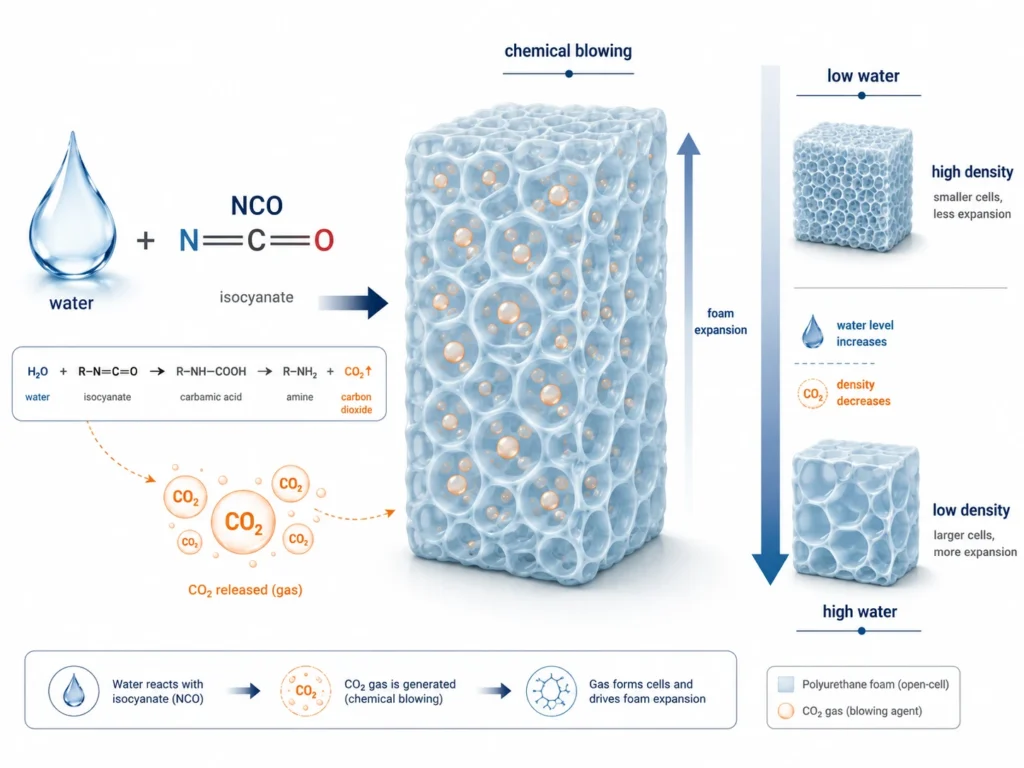
Role 1: Water as the Chemical Blowing Agent
The first role of water is to generate CO₂ gas, which physically expands the foam and creates its cellular structure.
When water reacts with isocyanate, the CO₂ released is trapped inside the rising polymer mass. As the polymer network builds and viscosity increases, those CO₂ bubbles cannot escape. They become foam cells.
The amount of CO₂ generated is directly proportional to the water level:
- More water → more CO₂ → more foam expansion → lower density.
- Less water → less CO₂ → less foam expansion → higher density.
This is the simplest way to see the relationship between water and foam density. In flexible slabstock and molded foam, water is the primary tool for controlling density.
| Water Level (parts per 100 polyol) | Typical Density Direction |
|---|---|
| Higher water (4.5+) | Lower density |
| Standard water (3.0–4.0) | Standard density |
| Lower water (2.5 or below) | Higher density |
The exact density at any water level depends on the full formulation — index, polyol grade, surfactant, catalyst — but the directional relationship is universal in water-blown foam.
This is the role most engineers think of when they hear “water in PU foam.” It is correct, but it is incomplete.
Role 2: Water as the Urea Network Builder
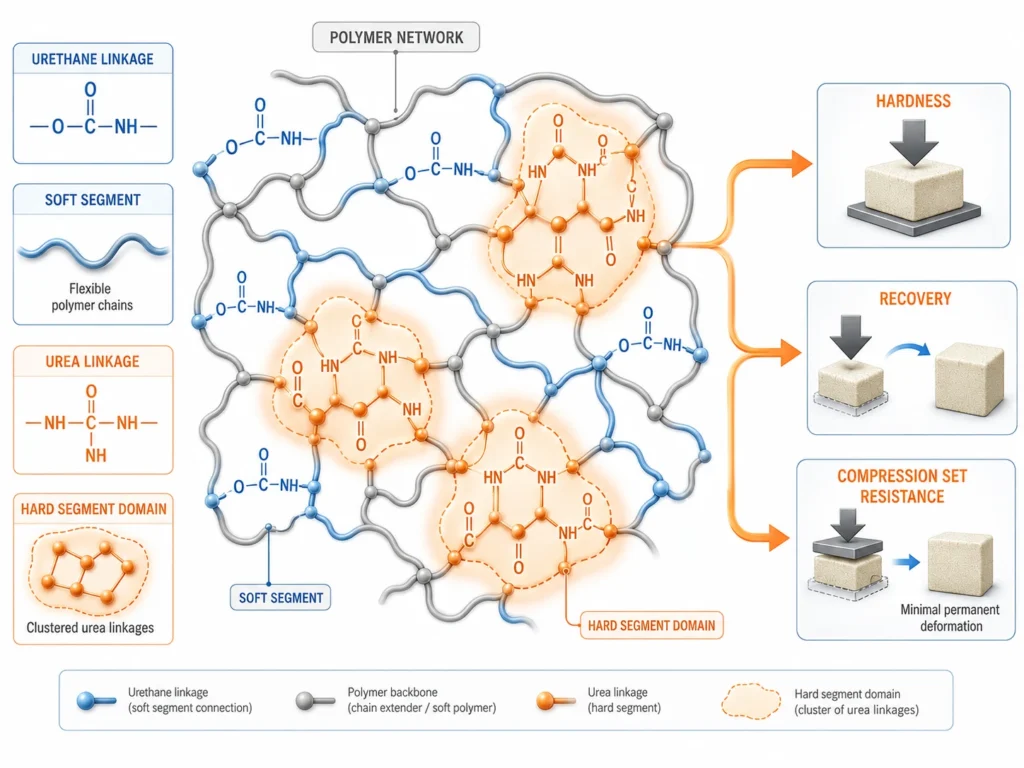
The second role of water is to build urea hard segments into the polymer network.
When water reacts with NCO in Step 1, it produces an amine intermediate. That amine is highly reactive — much more reactive than a polyol OH group. The amine reacts with another NCO group to form a urea linkage (R-NH-CO-NH-R’).
These urea linkages are different from the urethane linkages formed by polyol-NCO reactions:
| Linkage Type | Formed By | Role in Network |
|---|---|---|
| Urethane | Polyol OH + NCO | Soft / flexible segments |
| Urea | Water-derived amine + NCO | Hard segments |
Urea linkages have stronger hydrogen bonding than urethane linkages. They cluster together in the polymer network, forming hard segment domains — small, stiff regions inside the otherwise flexible foam.
These hard segment domains affect:
- Foam hardness and ILD
- Load-bearing behavior
- Compression set resistance
- Recovery after compression
- Long-term durability
- Resilience
More water → more urea → more hard segment formation → harder, firmer foam (within limits).
This is the part most engineers do not actively think about. When water level changes, two things happen at once: density changes (Role 1) and hard segment content changes (Role 2). That is why water adjustments produce more than just density effects.
Why Both Roles Happen Simultaneously
Water cannot perform Role 1 without also performing Role 2. The chemistry is sequential and unavoidable.
When water reacts with NCO, the carbamic acid intermediate decomposes immediately. The amine produced is highly reactive. In a polyurethane foam system with excess NCO available, that amine always reacts with another NCO to form urea — there is no way to stop it.
This means:
- Every CO₂ molecule released for blowing is paired with the formation of a urea linkage.
- Every part of water added to the formula increases both gas generation and hard segment content.
- Every part of water removed reduces both gas generation and hard segment content.
The two effects cannot be separated. A water adjustment is always a paired adjustment — it changes density and hardness simultaneously, and the changes go in the same direction.
| Water Change | Density Effect | Hard Segment Effect | Hardness Direction |
|---|---|---|---|
| Increase water | Lower density | More urea formation | Firmer (from hard segments) but lower density (from CO₂) |
| Decrease water | Higher density | Less urea formation | Softer (less hard segments) but higher density |
This is why water is so often misdiagnosed in foam troubleshooting. An engineer reduces water to firm up the foam, expecting hardness to rise from increased density. But the reduction in urea formation can offset the density gain, and the foam may not become as firm as expected.
The correct mental model is: water controls density AND hard segment content. Both move together, in the same direction, with every water change.
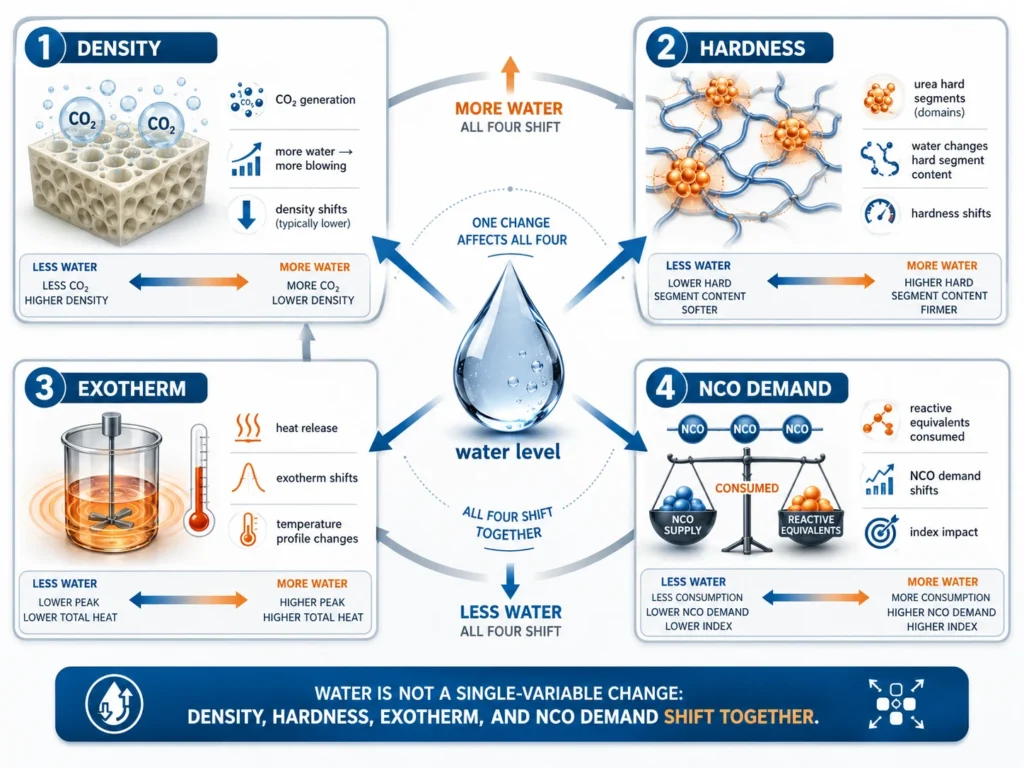
How Water Affects Four Properties at Once
The dual role of water means a single water adjustment can affect at least four foam properties simultaneously:
1. Density
CO₂ generation directly controls foam expansion. Higher water lowers density; lower water raises density.
2. Hardness / ILD
Urea hard segments contribute to load-bearing behavior. More water generally increases the hard segment fraction, which can raise hardness — but density falls at the same time, which lowers it. The net effect on hardness depends on the balance.
3. Exotherm
Both reactions (water + NCO → CO₂ + amine, and amine + NCO → urea) are highly exothermic. More water means more heat released during foam formation. In large blocks or thick parts, this can cause excessive internal temperature, leading to scorch (yellowing or burning of the foam core).
4. NCO Demand
Each part of water consumes 2 equivalents of NCO. Increasing water without increasing isocyanate parts will lower the actual running index. This affects cure, network completeness, and downstream foam properties.
| Water Change | Density | Hardness | Exotherm | NCO Demand |
|---|---|---|---|---|
| +1 part | ↓ | ↑ from urea, ↓ from density | ↑↑ | ↑ |
| −1 part | ↑ | ↓ from urea, ↑ from density | ↓↓ | ↓ |
No other component in flexible foam moves four properties simultaneously the way water does. That is what makes water the most powerful — and most dangerous — adjustment variable in the formula.
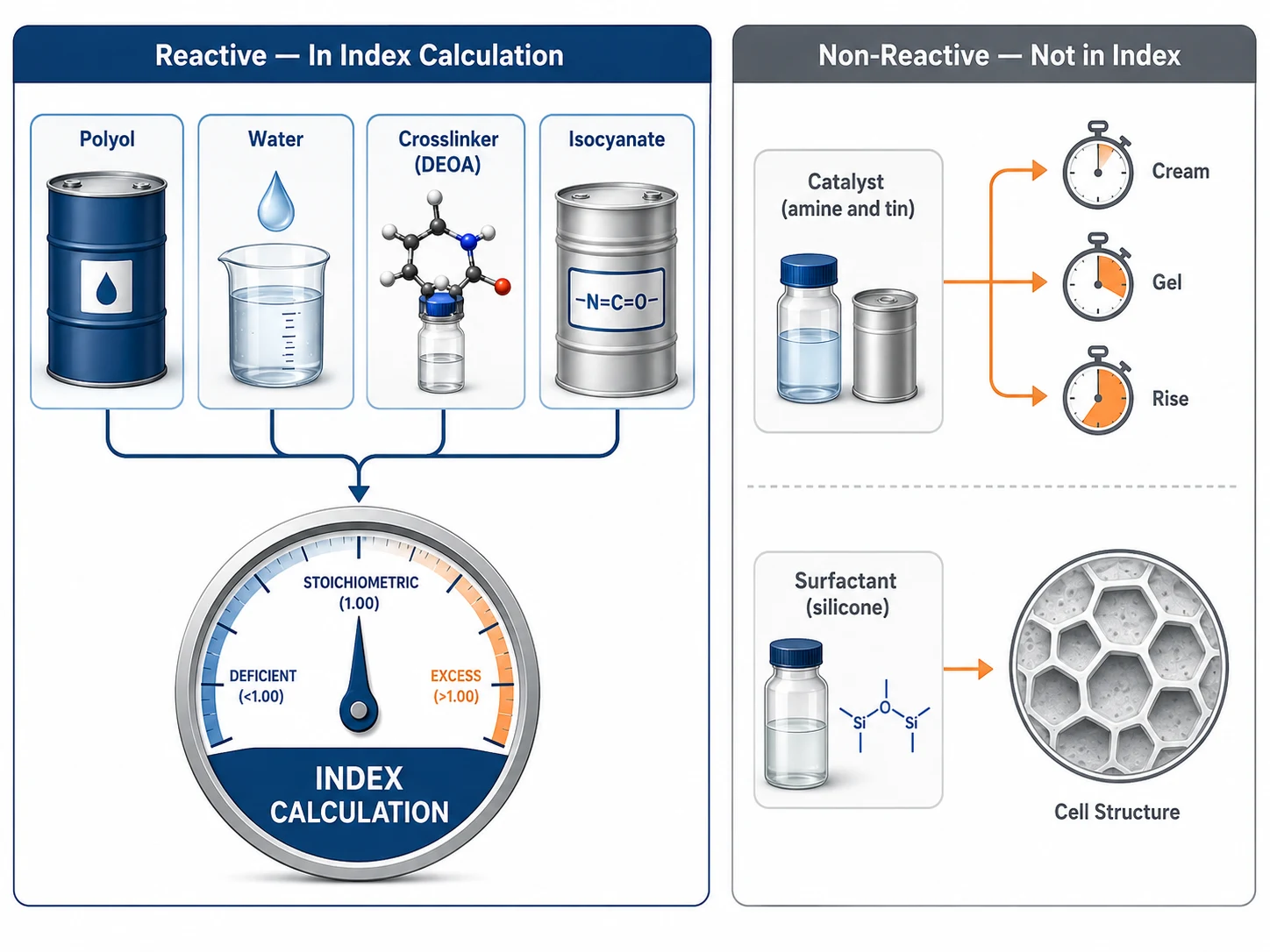
Water in the Index Calculation
Because water consumes two NCO equivalents per molecule, it must always be included in the isocyanate index calculation as a reactive hydrogen contributor.
Water equivalents in formula = water parts ÷ 9
For example, in a flexible foam formula with 4 parts of water:
Water equivalents = 4 ÷ 9 = 0.44444 equivalents
In a typical formula with 100 parts of polyol at OHV 51 and 0.5 parts of DEOA crosslinker, the reactive hydrogen breakdown looks like this:
| Component | Parts | EW (g/eq) | Equivalents | Equivalent % |
|---|---|---|---|---|
| Polyol | 100.00 | 1,100 | 0.09091 | 16.54% |
| Water | 4.00 | 9 | 0.44444 | 80.86% |
| DEOA | 0.50 | 35.0 | 0.01429 | 2.60% |
| Total reactive H | 0.54964 | 100% |
Water represents 2.56% of the formula by weight but 80.86% of the reactive hydrogen equivalents. That is a 31× difference between weight contribution and reactive contribution.
Implications for formulation control:
- A 0.3-part water increase looks tiny by mass but is a meaningful stoichiometric move.
- Every water change must be recalculated in the index.
- Water adjustments without isocyanate adjustments shift the actual running index.
- An apparent density correction may produce hardness, cure, and compression set side effects that look unrelated to water.
This is why water has to be treated as a reactive component first and a blowing agent second. The chemistry leads the physics, not the other way around.
What Goes Wrong When Water’s Dual Role Is Ignored
Many flexible foam quality problems trace back to water adjustments made without understanding the dual role. The common failure patterns are:
Density correction without index recalculation
The plant raises water to lower density. CO₂ generation increases as expected, density falls. But because water consumes NCO at two sites, more water also consumes more isocyanate equivalents. Without an isocyanate increase, the actual running index drops. The foam shows softer cure, weaker hardness, and possibly higher compression set — symptoms that look like a catalyst or crosslinker problem but are actually an index problem.
Hardness correction in the wrong direction
The plant reduces water to firm up soft foam, expecting hardness to rise from increased density. Density does rise, but the reduction in urea hard segments offsets the gain. Hardness moves less than expected, or in the opposite direction. The plant then increases catalyst or crosslinker to compensate, compounding the problem.
Scorch in large blocks
Water is highly exothermic. In large slabstock blocks or thick molded parts, increasing water also increases internal temperature. If the increase is significant, internal exotherm can exceed the safe range, causing yellow or brown discoloration in the core (scorch). The plant may blame catalyst or cure, but the root cause is excess water-driven exotherm.
Soft, low-recovery foam after a water reduction
The plant reduces water for cost or to raise density. Density rises, but urea content drops. The hard segment domains that supported recovery and compression set resistance are now weaker. The foam runs softer, less resilient, and with worse compression set than expected.
These failure patterns are not water mistakes alone — they are dual-role mistakes. Each one results from treating water as a blowing agent only, without accounting for its hard segment contribution and NCO consumption.

How to Manage Water Changes Correctly
Because water moves four properties simultaneously, every water-level adjustment should follow a structured workflow rather than being made in isolation.
- Define the target. What property is the water change trying to affect — density, hardness, or both?
- Predict all four effects. Before changing water, write down the expected impact on density, hardness (from urea), exotherm, and NCO demand.
- Recalculate the index. Use Water EW = 9 and verify whether the isocyanate level needs adjustment to maintain the target index.
- Adjust isocyanate if needed. If maintaining the index matters for the application, adjust isocyanate parts in proportion to the change in reactive hydrogen equivalents.
- Check exotherm risk. For large blocks or thick parts, verify that the new water level does not push internal temperature toward scorch.
- Review hard segment impact. For hardness-critical or compression-set-critical applications, consider whether the urea content change supports or fights the target.
- Run a controlled trial. Measure all four affected properties — density, hardness, recovery, and (where relevant) internal temperature.
- Document the change and outcome. Record the prediction and the actual result for future reference.
This workflow treats water as what it is: a reactive component with multiple connected effects, not a simple density dial.
Practical Reference: Water in Different Foam Types
The dual role of water applies everywhere water is used as a blowing agent, but the practical balance differs between foam types.
| Foam Type | Typical Water Level (parts) | Dual-Role Notes |
|---|---|---|
| Flexible slabstock | 3.0–5.0 | Water is the primary blowing agent; both roles are active and significant |
| Flexible molded | 2.5–4.5 | Lower water than slabstock; physical blowing agent may share blowing duty |
| HR foam | 3.0–4.5 | Hard segment role is critical for compression set performance |
| Rigid foam | 0.5–2.0 | Water is secondary; physical blowing agents (HFC, HFO, pentane) usually dominate |
| Spray foam | 0.5–2.0 | Water level controls density and exotherm at application; both roles active |
In all cases, the chemistry is the same: each water molecule consumes two NCO, releases one CO₂, and forms one urea linkage. The role does not change with foam type — only the amount of water and the relative importance of each effect changes.
Use the PolymerIQ Calculators
The dual role of water makes accurate index calculation essential whenever water level changes. The PolymersIQ NCO / TDI Index Calculator verifies whether a water adjustment requires an isocyanate adjustment to maintain the target index.
Open the NCO / TDI Index Calculator →
The PolymerIQ Equivalent Weight Calculator confirms that water EW is correctly entered as 9 g/eq alongside polyol EW from current OHV and isocyanate EW from current %NCO.
Open the Equivalent Weight Calculator →
The PolymerIQ Foam Density Estimator helps predict density impact before a water-level change reaches production. Use it together with the index calculator so density correction does not unintentionally drift the index.
Open the Foam Density Estimator →
For the article on how water level affects density, hardness, exotherm, and compression set in detail, read How Water Level Affects PU Foam Density, Hardness, Exotherm, and Compression Set.
For the article on common water adjustment mistakes, read 4 Water Adjustment Mistakes That Affect PU Foam Properties.
For why water’s equivalent weight is 9, read Why the Equivalent Weight of Water Is 9 in Polyurethane Foam.
For the technical article on which raw materials enter the index calculation, read Reactive vs Non-Reactive Components in PU Foam.
For the foundation article on isocyanate chemistry, read NCO Content in Isocyanate: What %NCO Means in PU Foam Formulation.
For the full equivalent weight guide, read Equivalent Weight in Polyurethane Foam: Complete Calculation Guide.
FAQs
What are the two roles of water in polyurethane foam?
Water has two reactive roles. First, it reacts with isocyanate to generate CO₂ gas, which expands the foam and creates the cellular structure (chemical blowing agent). Second, the same reaction produces an amine intermediate that reacts with another isocyanate to form urea linkages, which build hard segment domains in the polymer network. Both roles happen simultaneously — every part of water added increases both CO₂ generation and urea formation.
Why does water have an equivalent weight of 9 instead of 18?
Water’s molecular weight is 18 g/mol, but each water molecule consumes two NCO groups across the two-step reaction (one to form carbamic acid, then a second through the amine-to-urea step). Because water provides 2 reactive equivalents per molecule, equivalent weight = 18 ÷ 2 = 9 g/eq. Using EW = 18 in the formula sheet under-counts water’s reactive contribution by half and produces an incorrect index calculation.
How does water generate CO₂ in polyurethane foam?
When water reacts with an isocyanate group (NCO), it forms an unstable carbamic acid intermediate. The carbamic acid immediately decomposes into an amine and CO₂. The CO₂ is the gas that expands the foam. The amine then reacts with another NCO group to form a urea linkage, completing the two-step reaction.
What is a urea hard segment in polyurethane foam?
A urea hard segment is a region of urea linkages clustered together in the polymer network. Urea linkages have strong hydrogen bonding, so they tend to associate and form small, stiff domains within the otherwise flexible foam. These hard segments contribute to foam hardness, load-bearing behavior, recovery after compression, and compression set resistance. They are different from urethane linkages (formed by polyol OH + NCO), which create the soft flexible segments of the network.
Why does increasing water affect more than just density?
Because water has two reactive roles. Increasing water generates more CO₂ (lowering density) AND increases urea hard segment formation (raising hardness contribution) AND increases exotherm (raising heat release) AND consumes more NCO (lowering the running index unless isocyanate is also increased). A single water change moves at least four foam properties simultaneously, which is why water adjustments produce surprising side effects when only density is being targeted.
What happens if I increase water without adjusting isocyanate?
The actual running index drops. Each part of water added consumes 2 equivalents of NCO. If isocyanate parts stay constant, NCO equivalents are now divided by a larger reactive H denominator, so the index falls. The foam may become softer, undercured, or show compression set issues — symptoms that look unrelated to water but are actually caused by index drift. Every water-level change should trigger an index recalculation.
Why does reducing water sometimes not firm up the foam as expected?
Because water reduction has two opposing effects on hardness. Less water means less CO₂ and higher density, which generally raises hardness. But less water also means less urea formation and weaker hard segment domains, which lowers the hardness contribution from the urea network. The net effect depends on the balance — sometimes the density gain wins, sometimes the urea loss wins. This is one of the most common surprises in flexible foam troubleshooting.
Why is excess water dangerous in large foam blocks?
The water-isocyanate reaction is highly exothermic. More water means more heat released during foam formation. In large slabstock blocks or thick molded parts, the heat cannot escape from the core fast enough, and internal temperature can rise to levels that cause scorch (yellow or brown discoloration of the foam core). The risk increases with both water level and block thickness. For thick parts, water level should be balanced against acceptable internal exotherm, not just density target.
How much of the reactive hydrogen comes from water in flexible foam?
In a typical flexible slabstock formula with about 4 parts of water at EW = 9 and 100 parts of polyol at EW around 1,100, water contributes roughly 80% of the total reactive hydrogen equivalents. Polyol contributes about 16%, and crosslinker (e.g., DEOA) contributes the remaining 2–3%. This is why even a small water change can have a large stoichiometric effect — water dominates the reactive hydrogen side of the formula despite being only 2–3% by weight.
Does the dual role of water apply to rigid foam as well?
Yes, the chemistry is identical. Each water molecule still reacts with two NCO groups, releases CO₂, and forms a urea linkage. The difference in rigid foam is that water is usually present at lower levels (0.5–2.0 parts), and physical blowing agents (HFC, HFO, pentane) typically dominate density control. Water still contributes both CO₂ blowing and urea hard segments, but the relative importance of each effect is smaller compared to flexible foam where water is the primary blowing agent.
Key Takeaways
Water has two reactive roles in polyurethane foam, not one:
- Chemical blowing agent — water reacts with NCO to release CO₂, expanding the foam and reducing density.
- Urea network builder — the amine intermediate reacts with another NCO to form urea linkages, building hard segment domains in the polymer network.
These two roles happen simultaneously and cannot be separated. Every water adjustment produces both effects together.
The chemistry consumes two NCO groups per water molecule:
H₂O + 2 R-NCO → R-NH-CO-NH-R + CO₂
This gives water a very low equivalent weight:
Water EW = 18 ÷ 2 = 9 g/eq
Despite being only 2–3% of the formula by weight, water often contributes 70–85% of the reactive hydrogen equivalents in flexible foam. That is what makes water the most powerful adjustment variable.
A single water-level change moves four properties at once: density, hardness (from urea), exotherm, and NCO demand. This is why water adjustments produce surprising side effects when treated as a simple density dial.
Every water change must be paired with an index recalculation and, where the application requires it, an isocyanate adjustment. Treat water as a reactive component first, a blowing agent second.
Conclusion
If your flexible foam is showing unexplained hardness drift, compression set issues, scorch, or cure inconsistency after a density adjustment, the cause may be a water change that was treated as single-purpose when it was actually moving four properties at once.
PolymersIQ can help review your water level, urea hard segment contribution, exotherm risk, and index balance to identify whether the dual role of water has been correctly accounted for in your formulation.
To get accurate support, please share:
- Polyol grade, OHV, and supplier
- Isocyanate type and current CoA %NCO
- Current and previous water levels
- Recent water-level changes and the reasons they were made
- Target foam properties (density, hardness, compression set)
- Block size or part thickness (for exotherm review)
- Description of the foam quality issue and adjustments already tried
Contact PolymerIQ for a water and index review →