Introduction
The equivalent weight of water in polyurethane foam is 9, not 18.
This is one of the most important rules in PU foam formulation — and one of the most damaging mistakes when entered incorrectly.
Water has a molecular weight of 18 g/mol. Because of that, many engineers assume the equivalent weight of water is also 18. That assumption is wrong in polyurethane chemistry.
In PU foam, one water molecule ultimately consumes two NCO groups through the blowing reaction sequence. That is why the equivalent weight is calculated as:
Water EW = 18 ÷ 2 = 9 g/eq
If a formula spreadsheet uses 18 instead of 9, the water contribution is cut in half. The total reactive hydrogen equivalents become wrong. The calculated isocyanate demand becomes wrong. The index shown on the formula sheet no longer matches the chemistry in the reactor.
This article explains why water EW is 9, how the water-isocyanate reaction works, what happens when 18 is used by mistake, and how this error appears in foam production.
Why Water Equivalent Weight Is Not 18
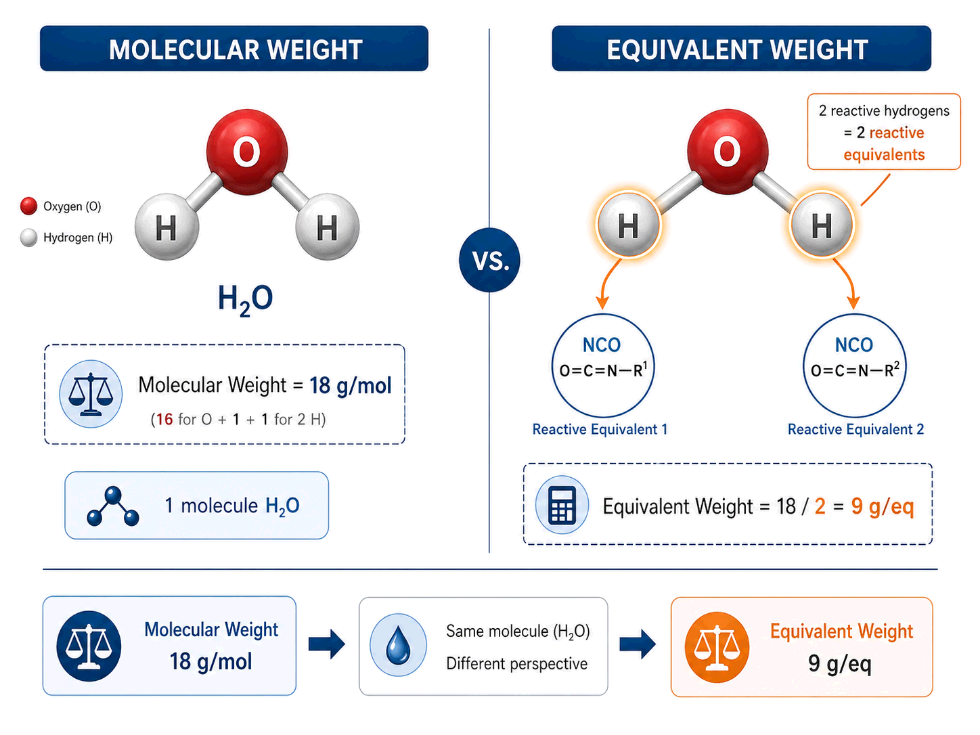
Water has a molecular weight of 18 g/mol. But equivalent weight is not always the same as molecular weight.
Equivalent weight means the mass of material that contains one equivalent of reactive functionality.
In polyurethane foam, water has two reactive hydrogens involved in the isocyanate reaction sequence. That means one mole of water provides two equivalents of reactivity toward NCO.
So the calculation is:
Water EW = Molecular Weight ÷ Reactive Hydrogen Count = 18 ÷ 2 = 9 g/eq
For polyurethane foam index calculation, the correct value is Water EW = 9, not 18.
Using 18 treats water as if it had only one reactive hydrogen. That cuts the water contribution in half and corrupts the index calculation.
How Water Reacts with Isocyanate in PU Foam
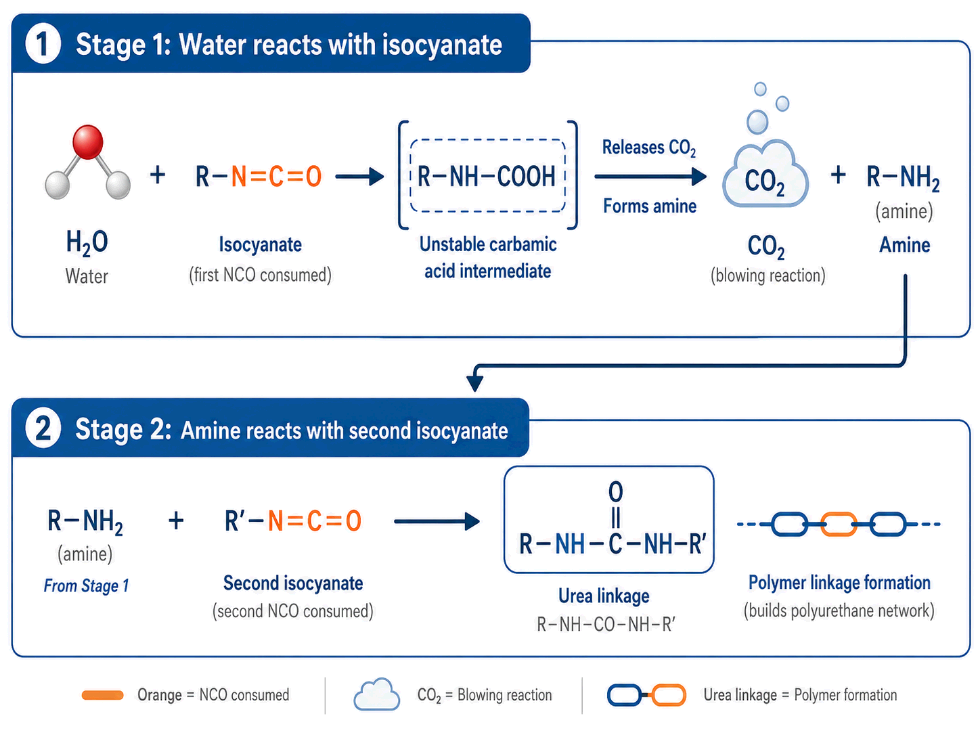
Water reacts with isocyanate in two main stages.
Stage 1: Water reacts with NCO. Water reacts with an isocyanate group to form an unstable carbamic acid intermediate. This intermediate quickly decomposes, releasing carbon dioxide (which blows the foam and forms cells) and producing a primary amine.
Stage 2: The amine reacts with another NCO group. The amine formed in Stage 1 is reactive. It reacts with a second isocyanate group to form a urea linkage.
This means one water molecule ultimately consumes two NCO groups:
- One NCO in the initial water reaction
- One NCO in the amine-to-urea reaction
This is the chemical reason water equivalent weight is 9. It is not an approximation or a rule of thumb — it comes directly from the reaction mechanism.
The Correct Water EW Calculation
The calculation is simple:
- Water molecular weight = 18 g/mol
- Reactive hydrogens = 2
- Water EW = 18 ÷ 2 = 9 g/eq
This means 9 grams of water contain one equivalent of reactive hydrogen functionality for the PU foam index calculation.
When calculating water equivalents in a formulation:
Water Equivalents = Water Parts ÷ 9
For example, if a flexible foam formula contains 4.0 parts water:
4.0 ÷ 9 = 0.44444 equivalents
If the formula uses EW = 18 instead:
4.0 ÷ 18 = 0.22222 equivalents
That is exactly half the correct value. The formula spreadsheet now believes there is much less reactive hydrogen demand than the chemistry actually has.

Worked Example: How EW Water = 18 Corrupts the Index
Let’s see how this mistake changes the full formula calculation.
Example flexible slabstock formula:
| Component | Parts | Correct EW | Correct Equiv. | Wrong EW | Wrong Equiv. |
|---|---|---|---|---|---|
| Polyol | 100 | 1,100 | 0.09091 | 1,100 | 0.09091 |
| Water | 4.0 | 9 | 0.44444 | 18 | 0.22222 |
| DEOA | 0.5 | 31 | 0.01613 | 31 | 0.01613 |
| Total H equiv. | 0.55148 | 0.32926 |
The correct total reactive hydrogen equivalents are 0.55148. Using water EW = 18 gives 0.32926.
Now assume the engineer targets Index 105 using the wrong equivalent system. NCO equivalents calculated from the wrong system:
0.32926 × 1.05 = 0.34572
But the actual correct reactive hydrogen equivalents are 0.55148. So the real running index is:
0.34572 ÷ 0.55148 × 100 = 62.7
The formula sheet says Index 105. The chemistry is running at approximately Index 62.7.
This is not a small error. It is a completely wrong stoichiometric foundation.

What This Error Looks Like in Production
A water equivalent weight error does not always create a dramatic visual failure. The foam may still rise. The block may still form. Operators may not immediately see the problem at the machine.
But the properties can be seriously wrong.
If water is entered as 18 instead of 9 and the isocyanate quantity is calculated from that wrong value, the foam can be severely under-indexed.
Common symptoms include:
- Softer foam than expected
- ILD below target
- Poor compression set
- Weak recovery
- Slower or weaker cure
- Tacky feel during early cure
- Poor aging performance
- Customer complaints after use
- Confusing response to catalyst adjustments
This kind of problem can be difficult to diagnose because it looks like a process issue. The team may adjust catalyst, silicone, cure temperature, water level, or crosslinker dosage. Some changes may improve one symptom temporarily. But the root cause remains inside the calculation.
The spreadsheet must be checked.

Why This Mistake Stays Hidden
The water EW mistake stays hidden because the formula sheet often looks internally consistent.
The numbers may be formatted correctly. The index cell may show the target value. The spreadsheet may have been used for years.
But the spreadsheet is only as accurate as the assumptions inside it. If water EW is entered as 18, every downstream calculation built on that value becomes wrong.
This mistake is especially common in legacy formulas because:
- Water molecular weight is commonly remembered as 18
- Engineers may copy old spreadsheets without checking the chemistry
- The formula may have been empirically adjusted over time
- Production teams may trust a formula because it has been used for years
- Troubleshooting often focuses on machine and process variables first
- The equivalent weight layer is rarely audited
This is why a formula can carry the same error for months or years. The plant may keep adding practical corrections on top of a wrong calculation foundation. That creates a formula that works only by accident — and becomes difficult to transfer, scale, or troubleshoot.
How to Check Your Formula Today
Checking for this mistake is simple.
Open your formula sheet and find the equivalent weight value used for water. It should be 9, not 18.
Then check how the water equivalents are calculated:
- Correct: Water equivalents = Water parts ÷ 9 (e.g., 4.0 ÷ 9 = 0.44444)
- Wrong: Water equivalents = Water parts ÷ 18 (e.g., 4.0 ÷ 18 = 0.22222)
After correcting the water equivalent weight, the full index must be recalculated. Do not change only the water EW cell and assume the formula is now production-ready. The isocyanate quantity may also need to be recalculated based on the correct total reactive hydrogen equivalents and target index.
A safe review should include:
- Confirm water EW = 9
- Confirm polyol EW from actual OHV
- Confirm isocyanate EW from actual %NCO
- Confirm all crosslinkers and chain extenders are included
- Recalculate total reactive hydrogen equivalents
- Recalculate required NCO equivalents
- Recalculate TDI or MDI parts
- Compare the corrected formula against current production results

Use the PolymerIQ Isocyanate Index Calculator
Manual calculation is important because engineers should understand why water EW is 9. But in production, the calculation must also be checked quickly and consistently.
The PolymersIQ Isocyanate Index Calculator can help verify whether your formula is using the correct equivalent weights and delivering the intended index.
Use it to check water equivalent weight, total reactive hydrogen equivalents, required NCO equivalents, TDI or MDI parts, actual running index, and the effect of correcting EW errors.
Open the Isocyanate Index Calculator →
For the complete equivalent weight calculation guide, read Equivalent Weight in Polyurethane Foam: Complete Calculation Guide.
For common production spreadsheet mistakes, read 5 Equivalent Weight Mistakes That Damage PU Foam Production.
For the full isocyanate index calculation method, read Isocyanate Index Calculation Guide for PU Foam Engineers.
FAQs
Why is the equivalent weight of water 9 and not 18?
Water has a molecular weight of 18 g/mol, but each water molecule has two reactive hydrogens and consumes two NCO groups during the blowing reaction. So the equivalent weight is 18 ÷ 2 = 9 g/eq. Equivalent weight measures the mass per reactive equivalent — not the mass per molecule — so the divisor matters.
How does water actually consume two NCO groups?
The reaction happens in two stages. First, water reacts with one NCO group to form an unstable carbamic acid that releases CO₂ (the blowing gas) and forms a primary amine. Second, the amine reacts with another NCO group to form a urea linkage. The result: one water molecule consumes two NCO groups.
What happens if I use water EW = 18 by mistake?
Using 18 cuts the calculated water equivalents in half. The total reactive hydrogen equivalents become wrong, the calculated isocyanate demand becomes wrong, and the actual running index can be much lower than the formula sheet shows. The foam may still rise but will likely be under-indexed.
What does under-indexed foam look like in production?
Common symptoms include softer foam than expected, ILD below target, poor compression set, weak recovery, slower or weaker cure, tacky feel during early cure, and poor aging performance. The foam may rise normally, which is why this error often stays hidden for a long time.
Can this error explain unexplained compression set failures?
Yes. If water EW is wrong and the foam is under-indexed, crosslink density is lower than designed, which directly affects compression set, recovery, and aging stability. Compression set problems that don’t respond to catalyst or silicone changes should trigger an EW audit.
How do I check if my formula has this mistake?
Open your formula sheet and find the equivalent weight value used for water. If it shows 18 instead of 9, the calculation is wrong. Also check the equivalents formula: it should be water parts ÷ 9, not water parts ÷ 18.
Should I just change the water EW cell from 18 to 9?
No — that alone is not enough. After correcting water EW, the entire index must be recalculated, and the isocyanate quantity may need to change as well. Changing only the water EW cell without recalculating the rest of the formula may create new imbalances.
Why has this mistake stayed in some formula sheets for years?
Because the formula sheet looks internally consistent. The index cell shows the target value, the math is formatted correctly, and the spreadsheet has been used for a long time. Troubleshooting usually focuses on machines and process variables, and the equivalent weight layer is rarely audited.
Does this rule apply to flexible foam, rigid foam, and elastomers?
Yes. Water has the same chemistry — two reactive hydrogens, two NCO groups consumed — regardless of the polyurethane system. Water EW = 9 applies to flexible slabstock, HR foam, rigid foam, elastomers, and any PU system that uses water as a blowing agent or reactive component.
Does the water purity or temperature change the equivalent weight?
No. The equivalent weight comes from the reaction stoichiometry, not from physical conditions. As long as the water is participating in the standard PU blowing reaction, EW = 9 is the correct value to use.
Key Takeaways
The equivalent weight of water in polyurethane foam is 9, not 18.
Water has a molecular weight of 18, but it has two reactive hydrogens involved in the isocyanate reaction sequence:
Water EW = 18 ÷ 2 = 9 g/eq
Using 18 instead of 9 cuts the calculated water equivalents in half. This can severely corrupt the isocyanate index calculation and cause the actual running index to be much lower than the formula sheet suggests.
The foam may still rise and look normal, but it can show soft hardness, poor compression set, weak recovery, and confusing production behaviour.
Every PU foam formulation spreadsheet should be checked to confirm that water equivalent weight is entered as 9. A single wrong number can silently damage the entire stoichiometric calculation.
Conclusion
If your foam is consistently soft, failing compression set, or responding unpredictably to catalyst and process adjustments, the problem may not be the machine.
It may be the equivalent weight foundation inside the formula sheet.
PolymersIQ can help audit your formulation, verify water EW, recalculate the true index, and identify whether a hidden stoichiometric error is affecting production.
To get accurate support, please share:
- A screenshot or copy of your current formula sheet (with EW values)
- Polyol OHV and isocyanate %NCO values currently in use
- Water level and any crosslinkers or chain extenders
- Target index and actual foam properties (ILD, compression set, density)
- Description of the production issue you are facing
Contact PolymerIQ for a stoichiometric formulation audit →

