Introduction
Equivalent weight is one of the most important calculation values in polyurethane foam formulation.
It is also one of the most common sources of hidden formulation errors.
A foam formula can look correct on paper. The index may appear correct. The raw material parts may look familiar. The production team may check catalysts, silicone, temperature, density, and machine settings. But if even one equivalent weight value is wrong, the entire stoichiometric balance can be wrong.
This is why equivalent weight matters.
Equivalent weight is the value that connects raw material data to polyurethane chemistry. It converts each reactive component into a common basis so the formulator can calculate isocyanate demand correctly.
Polyol, isocyanate, water, and crosslinkers all have different structures and different reactive groups. Equivalent weight allows all of them to be compared on the same chemical basis.
This guide explains what equivalent weight means, how it differs from molecular weight, and how to calculate equivalent weight for every major PU foam component.
What Is Equivalent Weight?
Equivalent weight answers one simple question:
How many grams of this material contain one equivalent of reactive groups?
In polyurethane formulation, equivalent weight is not just a theoretical value. It is the foundation of stoichiometric balance. It tells the formulator how much of a material is required to provide one mole-equivalent of reactive functionality.
For example:
- Polyol provides hydroxyl groups.
- Isocyanate provides NCO groups.
- Water provides reactive hydrogens.
- Crosslinkers provide hydroxyl, amine, or other active hydrogen groups.
Each of these materials has a different molecular weight and a different number of reactive groups. Equivalent weight normalizes them so they can be used in the same calculation system.
Without equivalent weight, the isocyanate index calculation has no reliable foundation.
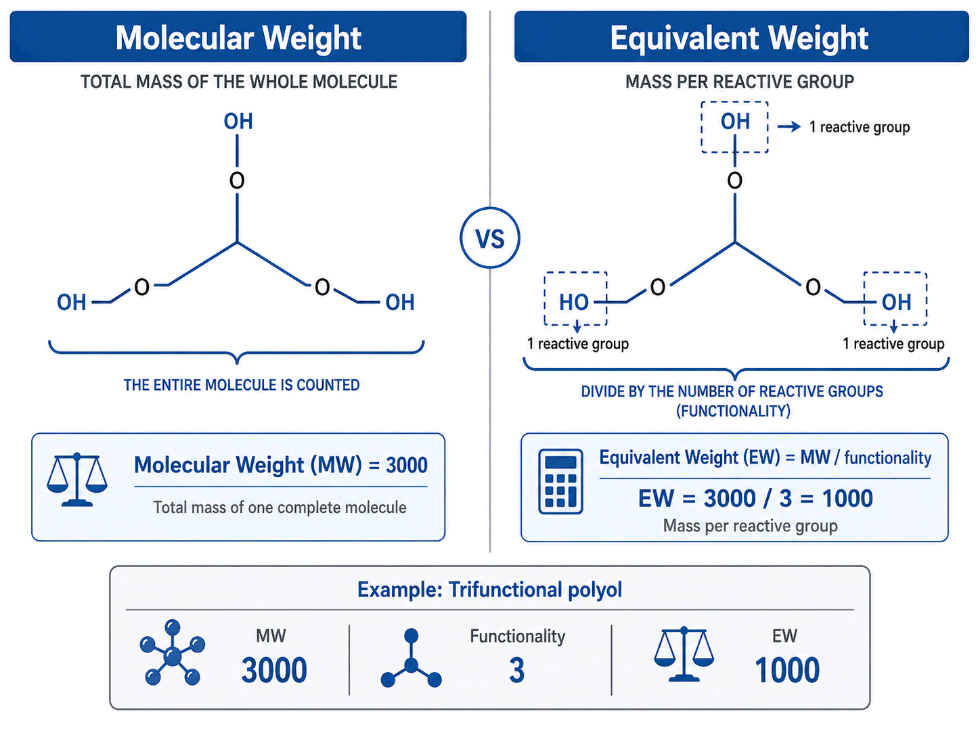
Equivalent Weight vs Molecular Weight
A common mistake is confusing equivalent weight with molecular weight. They are not always the same.
- Molecular weight is the mass of one mole of complete molecules.
- Equivalent weight is the mass that contains one mole-equivalent of reactive groups.
For a monofunctional material, molecular weight and equivalent weight can be the same. But for materials with more than one reactive group, equivalent weight is lower than molecular weight.
The general relationship is:
Equivalent Weight = Molecular Weight ÷ Functionality
For example, a trifunctional polyol with molecular weight 3,000 g/mol has three reactive hydroxyl groups per molecule.
So:
EW = 3,000 ÷ 3 = 1,000 g/eq
This means 1,000 grams of that polyol contains one equivalent of hydroxyl reactivity.
The same principle explains why water has an equivalent weight of 9, not 18. Water has a molecular weight of 18, but it has two reactive hydrogens involved in the isocyanate reaction.
So:
EW water = 18 ÷ 2 = 9 g/eq
This distinction is critical. A formulation that uses molecular weight where equivalent weight is required can produce a completely wrong index calculation.
Why Equivalent Weight Matters in PU Foam Formulation
Polyurethane foam chemistry is based on the reaction between isocyanate groups and active hydrogen groups.
The key reaction balance is:
- NCO groups from isocyanate
- OH groups from polyol
- Reactive hydrogens from water
- Reactive groups from crosslinkers or chain extenders
The isocyanate index depends on these equivalent relationships.
If the equivalent weight of one component is wrong, the calculated number of reactive equivalents is wrong. If the reactive equivalents are wrong, the isocyanate requirement is wrong. If the isocyanate requirement is wrong, the actual foam properties can shift.
This can affect:
- Foam hardness
- Compression set
- Resilience
- Crosslink density
- Cure behaviour
- Aging stability
- Batch-to-batch consistency
Equivalent weight errors are dangerous because the foam may still rise and look normal. The problem usually appears later in physical testing or customer use.
How to Calculate Polyol Equivalent Weight
For polyols, equivalent weight is calculated from hydroxyl value.
The formula is:
Polyol EW = 56,100 ÷ OHV
Where:
- EW = equivalent weight in g/eq
- OHV = hydroxyl value in mg KOH/g
- 56,100 = conversion constant from the KOH titration basis
The constant 56,100 comes from the molecular weight of potassium hydroxide (56.1 g/mol) multiplied by 1,000 for unit conversion.
Example
If a polyol has an OHV of 51 mg KOH/g:
EW = 56,100 ÷ 51 = 1,100 g/eq
So a polyol with OHV 51 has an equivalent weight of approximately 1,100 g/eq. This means 1,100 grams of that polyol contains one equivalent of reactive hydroxyl groups.
This calculation should be done using the actual OHV from the Certificate of Analysis, not only the nominal value from the Technical Data Sheet.

How to Calculate Isocyanate Equivalent Weight
For isocyanates, equivalent weight is calculated from the percentage of NCO.
The formula is:
Isocyanate EW = 4,200 ÷ %NCO
Where:
- EW = equivalent weight in g/eq
- %NCO = actual NCO percentage from the Certificate of Analysis
- 4,200 = molecular weight of the NCO group (42 g/mol) multiplied by 100
Example 1: TDI 80/20
If TDI has a %NCO of 48.3:
EW = 4,200 ÷ 48.3 = 86.96 g/eq
So the TDI equivalent weight is approximately 87 g/eq.
Example 2: MDI
If MDI has a %NCO of 31.5:
EW = 4,200 ÷ 31.5 = 133.33 g/eq
So the MDI equivalent weight is approximately 133 g/eq.
The same formula applies to TDI, MDI, polymeric MDI, and modified isocyanates. The constant does not change. The variable is the actual %NCO value.
For production calculation, use the %NCO from the Certificate of Analysis, not only the general TDS range.

How to Calculate Water Equivalent Weight
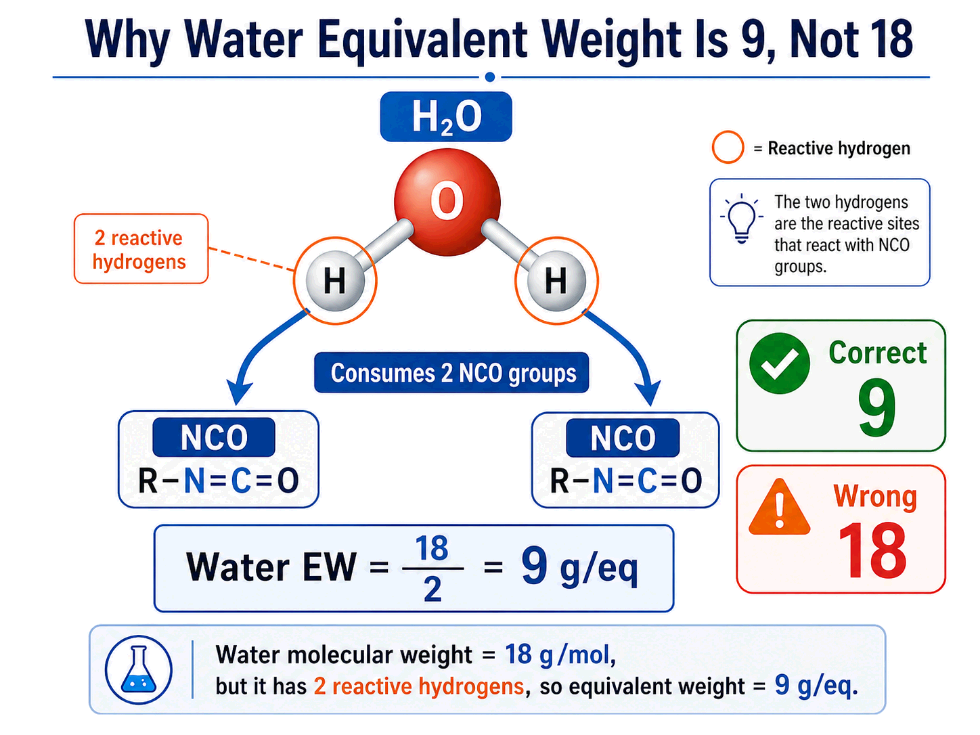
Water is one of the most important components in flexible polyurethane foam formulation. It is also one of the easiest to calculate incorrectly.
Water has a molecular weight of 18 g/mol. But its equivalent weight in polyurethane formulation is not 18.
Water has two reactive hydrogens involved in the isocyanate reaction sequence. One water molecule consumes two NCO groups.
Therefore:
Water EW = 18 ÷ 2 = 9 g/eq
This value is fixed.
For PU foam index calculation: water equivalent weight is 9, not 18.
Using 18 instead of 9 cuts the calculated water contribution in half and can severely distort the isocyanate index calculation.
The detailed water equivalent weight error and its production consequences are covered in a separate article — the water EW mistake is one of the most damaging single-number errors in PU foam formulation.
How to Calculate Crosslinker Equivalent Weight
Crosslinkers and chain extenders must also be included in equivalent weight calculations if they contain reactive groups.
For hydroxyl-based crosslinkers, the same formula used for polyols can often be applied:
Crosslinker EW = 56,100 ÷ OHV
Example: Glycerol
If glycerol has an OHV of approximately 1,827 mg KOH/g:
EW = 56,100 ÷ 1,827 = 30.7 g/eq
So the equivalent weight is approximately 31 g/eq.
This is much lower than the equivalent weight of a typical flexible foam polyol. That means even small quantities of crosslinker can contribute meaningful reactive equivalents.
Important note about amine-functional crosslinkers
Some crosslinkers or chain extenders contain more than hydroxyl groups. For example, some amine-functional materials include reactive amine hydrogens as well. In those cases, an OHV-only calculation may not capture all reactive functionality.
The correct approach is to account for all active hydrogen groups that react with isocyanate.
This topic is covered in more depth in a separate article on equivalent weight mistakes, because missing reactive groups in crosslinkers can quietly distort index and network structure.

Complete Equivalent Weight Reference Table
The table below summarizes the main equivalent weight formulas used in PU foam formulation.
| Component | EW Formula | Key Variable | Worked Example |
|---|---|---|---|
| Polyol | 56,100 ÷ OHV | OHV from CoA | OHV 51 → EW 1,100 |
| Isocyanate | 4,200 ÷ %NCO | %NCO from CoA | 48.3% NCO → EW 86.96 |
| Water | 18 ÷ 2 | Fixed value | EW = 9 |
| Hydroxyl crosslinker | 56,100 ÷ OHV | OHV of crosslinker | OHV 1,827 → EW 30.7 |
Every number in this table can feed into the isocyanate index calculation.
If one EW value is wrong, the index becomes unreliable. If multiple EW values are wrong, the production symptoms can become confusing and difficult to diagnose.
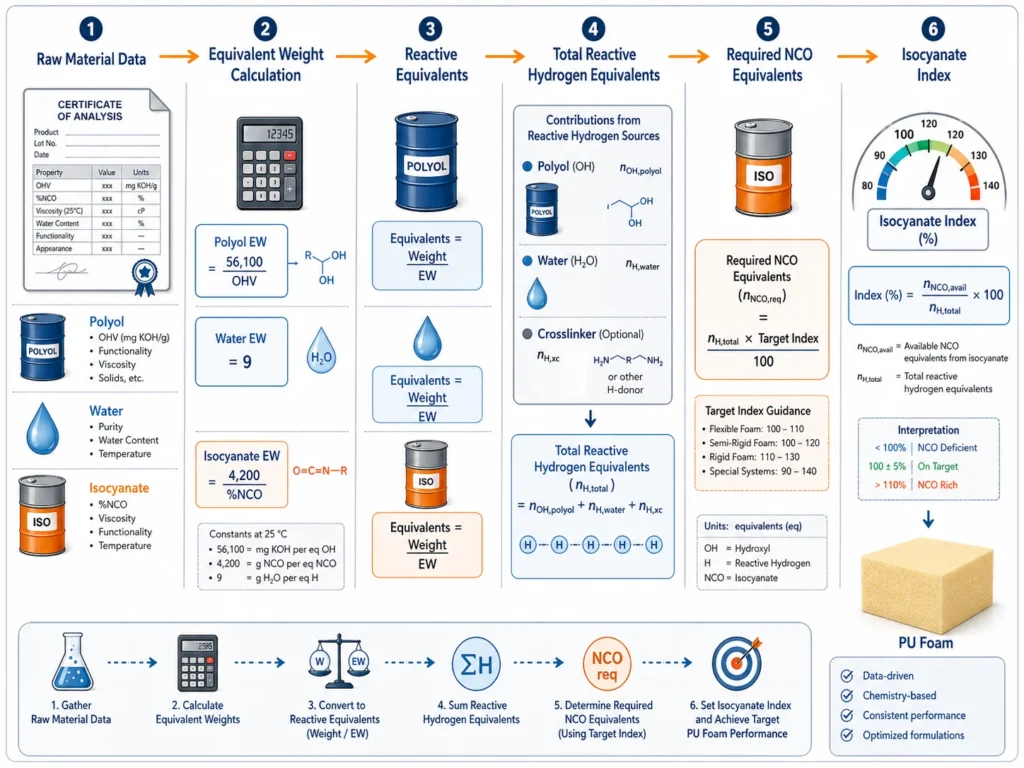
How Equivalent Weight Feeds Into Isocyanate Index
Equivalent weight is used to calculate the number of reactive equivalents in the formula.
The general formula is:
Reactive Equivalents = Parts by Weight ÷ Equivalent Weight
For example, if a formulation contains 100 parts of polyol with EW 1,100:
Polyol equivalents = 100 ÷ 1,100 = 0.09091
If the formula contains 4 parts of water with EW 9:
Water equivalents = 4 ÷ 9 = 0.44444
Each reactive component is converted into equivalents. Then all reactive hydrogen equivalents are added together. The isocyanate required is calculated from that total and the target index.
This is why equivalent weight is not an isolated calculation. It is part of the full stoichiometric system.
Wrong EW → wrong equivalents → wrong index → wrong foam properties.
Practical Rules for Equivalent Weight Calculation
Use these rules to avoid common formulation mistakes:
- Do not confuse molecular weight with equivalent weight. Molecular weight describes the whole molecule. Equivalent weight describes the mass per reactive group.
- Use actual CoA values when available. Polyol OHV and isocyanate %NCO can vary by batch.
- Use water EW = 9. Water has two reactive hydrogens and consumes two NCO groups.
- Recalculate EW when OHV changes. Polyol equivalent weight is not fixed if OHV changes.
- Recalculate isocyanate EW when %NCO changes. The isocyanate equivalent weight depends on actual %NCO.
- Include crosslinkers and chain extenders. Any reactive component must be included in the stoichiometric calculation.
- Check all active hydrogens. Some materials contain amine groups or other reactive functionality not captured by simple OHV alone.
- Audit old formula sheets. Legacy spreadsheets often contain copied EW values that may no longer match current raw material data.
Use the PolymerIQ Equivalent Weight Calculator
Manual calculation is useful because every foam engineer should understand the chemistry behind equivalent weight. But in production, the calculation must also be fast and consistent.
The PolymersIQ Equivalent Weight Calculator helps you calculate equivalent weight from OHV quickly.
Use it when:
- A new polyol batch arrives
- The CoA OHV is different from the design value
- You are checking a formulation before production
- You are preparing an isocyanate index calculation
- You are auditing an old formula sheet
Open the Equivalent Weight Calculator →
For a deeper article on the water calculation error, read Why the Equivalent Weight of Water Is 9 in Polyurethane Foam.
For common production mistakes, read 5 Equivalent Weight Mistakes That Damage PU Foam Production.
For the full isocyanate index method, read Isocyanate Index Calculation Guide for PU Foam Engineers.
FAQs
What is equivalent weight in polyurethane foam formulation?
Equivalent weight is the mass of material that contains one mole-equivalent of reactive groups. In polyurethane foam, it is used to convert each reactive component (polyol, isocyanate, water, crosslinker) into a common basis so the formulator can calculate isocyanate demand and index correctly.
How is equivalent weight different from molecular weight?
Molecular weight is the mass of one mole of complete molecules. Equivalent weight is the mass per reactive group. For monofunctional materials they can be the same, but for multifunctional materials, equivalent weight is lower than molecular weight. The relationship is EW = Molecular Weight ÷ Functionality.
How do I calculate polyol equivalent weight?
Use EW = 56,100 ÷ OHV, where OHV is the hydroxyl value in mg KOH/g. The constant 56,100 comes from the molecular weight of potassium hydroxide (56.1 g/mol) multiplied by 1,000 for unit conversion. Always use the actual OHV from the Certificate of Analysis, not the nominal TDS value.
How do I calculate isocyanate equivalent weight?
Use EW = 4,200 ÷ %NCO, where %NCO is the percentage of NCO groups by weight. The constant 4,200 comes from the NCO group molecular weight (42 g/mol) multiplied by 100. The same formula applies to TDI, MDI, polymeric MDI, and modified isocyanates — only the %NCO value changes.
Why is the equivalent weight of water 9 and not 18?
Water has a molecular weight of 18, but each water molecule has two reactive hydrogens and consumes two NCO groups during the blowing reaction. So the equivalent weight is 18 ÷ 2 = 9 g/eq. Using 18 instead of 9 cuts the calculated water contribution in half and severely distorts the isocyanate index.
Do I need to calculate equivalent weight for crosslinkers?
Yes. Hydroxyl-based crosslinkers use the same formula as polyols (EW = 56,100 ÷ OHV). Glycerol, for example, has an OHV around 1,827 mg KOH/g, giving an EW of about 31 g/eq. Because crosslinker EW is much lower than polyol EW, even small amounts contribute meaningful reactive equivalents to the calculation.
What about amine-functional crosslinkers and chain extenders?
Materials with amine groups or other active hydrogens cannot be captured by an OHV-only calculation. The correct approach is to account for all active hydrogen groups that react with isocyanate. Missing reactive groups in crosslinkers can silently distort the index and the polymer network.
How does equivalent weight feed into the isocyanate index?
Reactive equivalents are calculated as Parts ÷ Equivalent Weight for each component. All reactive hydrogen equivalents are summed, then multiplied by the target index to determine required NCO equivalents. The isocyanate quantity is then calculated as Required NCO equivalents × Isocyanate EW. Wrong EW values create wrong equivalents and wrong index.
Should I recalculate equivalent weight when raw material batches change?
Yes. Polyol EW changes when OHV changes. Isocyanate EW changes when %NCO changes. Treating EW as a fixed value copied from an old formula sheet is one of the most common causes of hidden formulation drift.
What’s the most common equivalent weight mistake in PU foam formulation?
Using water EW as 18 instead of 9. Because water is usually one of the largest contributors to reactive hydrogen equivalents in flexible foam, getting this single value wrong can shift the running index by many points and produce foam that is significantly harder than expected.
Key Takeaways
Equivalent weight is the mass of material that contains one equivalent of reactive groups. It is not always the same as molecular weight.
In polyurethane foam formulation, equivalent weight is needed for every reactive component because the isocyanate index depends on reactive equivalents.
The main formulas are:
- Polyol EW = 56,100 ÷ OHV
- Isocyanate EW = 4,200 ÷ %NCO
- Water EW = 18 ÷ 2 = 9
- Hydroxyl crosslinker EW = 56,100 ÷ OHV
Equivalent weight should be treated as a live calculation, not a fixed value copied from an old formula sheet.
- If OHV changes, polyol EW changes.
- If %NCO changes, isocyanate EW changes.
- If water is entered as 18 instead of 9, the index calculation becomes seriously wrong.
A correct equivalent weight system is the foundation of a correct isocyanate index calculation.
Conclusion
If your foam formula has been adjusted many times over the years, the equivalent weight values in the spreadsheet may no longer be correct.
PolymersIQ can help review your formulation, check every equivalent weight value, and identify whether hidden stoichiometric errors are affecting foam quality.
To get accurate support, please share:
- Polyol grade, OHV, and supplier
- Isocyanate type and %NCO from the Certificate of Analysis
- Water level and any crosslinkers or chain extenders in use
- Current EW values used in the formula sheet
- Description of the foam quality issue (if any)
Contact PolymerIQ for a stoichiometric formulation audit →

